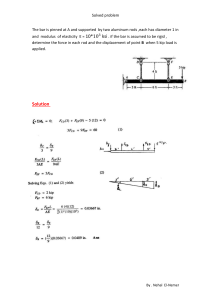
Copyright # 2016 ASM InternationalW All rights reserved www.asminternational.org ASM Handbook, Volume 4E, Heat Treating of Nonferrous Alloys G.E. Totten and D.S. MacKenzie, editors Quenching of Aluminum Alloys D. Scott MacKenzie, Houghton International Niels Bogh, Bogh Industries Tom Croucher, Tom Croucher and Associates QUENCHING refers to the rapid cooling of metal from the solution treating temperature, typically between 465 and 565 C (870 and 1050 F) for aluminum alloys. The fundamental objective of quenching is to preserve, as nearly as possible, a metastable solid solution formed at the solution heat treating temperature, by rapidly cooling to some lower temperature, usually near room temperature. When quenching rates from the solution temperature are sufficiently rapid to retain a solid solution, solute atoms are available to form zones of homogeneous (coherent or semicoherent) precipitation for strengthening by age hardening at room temperature or moderately elevated temperatures. In addition, another purpose of quenching is to maintain a certain minimum number of vacant lattice sites to assist in promoting the low-temperature diffusion during the aging stage of precipitation hardening. Precipitation during quenching can lead to localized overaging, a loss of grain-boundary corrosion resistance, and, in extreme cases, poor response during the age hardening treatment. When quenching rates from the solution temperature are not sufficiently rapid, the solute atoms that diffuse to grain boundaries, as well as the vacancies that migrate (with extreme rapidity) to disordered regions, are irretrievably lost for practical purposes and fail to contribute to the process of age hardening. The highest attainable strengths from aging hardening are generally associated with the most rapid quenching rates. Nonetheless, a maximum quenching rate is not necessarily a one-sided argument, because both distortion and residual stresses also develop with an increase in the rate of cooling. A balance must be obtained between the need to quench sufficiently fast to retain most of the hardening elements and compounds in solution and the need to minimize residual stress and distortion in the parts being quenched. Obtaining properties and low distortion is usually a balancing act. Often, optimal properties are obtained at the expense of high residual stresses or high distortion. Low distortion or residual stresses are usually obtained at a sacrifice in properties. Therefore, the optimum rate of cooling is one where properties are just met, so that distortion is reduced. Aluminum also is extremely prone to distortion due to both solution heat treatment and quenching. The coefficient of linear expansion of aluminum is twice that of steel (2.38 105 mm/mm C for aluminum versus 1.12 105 mm/mm C for steel), and large thermal strains can develop within and across the surface of parts due to thermal expansion during solution heat treatment. In addition, temperatures during solution treatment approach the liquidus temperature and result in low strength and high plasticity. The most troublesome changes in dimensions and shape can occur during quenching, due to the extent of nonuniform cooling, particularly in thin sections of parts that contain variations in thickness. If distortion is to be minimized, the temperature differences between different areas of a part must be minimized consistent with cooling all regions of the part sufficiently fast to avoid excessive precipitation during quenching. Aluminum alloys have a relatively high thermal conductivity, between 1.4 and 2.38 W/cm K (975 and 1650 Btu in./h ft2 F), compared to a conductivity of approximately 0.14 to 0.29 W/cm K (100 to 200 Btu in./h ft2 F) for austenite in most carbon and lowalloy steels. The high conductivity of aluminum can be both a benefit and a problem. If heat is being rapidly extracted at the part surface by the quenchant, the high conductivity results in rapid temperature losses in thin sections and large temperature differences between thick and thin sections. If heat is being extracted more slowly, the high metal conductivity aids in maintaining temperature uniformity within the part. This article provides an overview on the factors used to determine a suitable cooling rate and the appropriate quenching process to develop a suitable cooling rate. The practical difficulty lies in establishing just how fast a part of a particular alloy must be quenched, while also minimizing, as much as possible, thermal gradients within the part that may cause plastic deformation and residual stress. To retain a sufficient amount of alloying elements in solid solution for subsequent age hardening, it is necessary to determine a suitable cooling rate for a given alloy and part by considering the following questions (Ref 1): Are there some temperatures during the quench when diffusion-controlled reactions will occur faster? What is the critical quench rate for how fast the part must be quenched to effectively retard diffusion of solute atoms in the solid solution? What happens when cooling rates during a quench are slower than the critical quench rate? When a suitable cooling rate is determined, the appropriate quench process is determined by several operational factors, such as: The heat-extracting potential of the quench- ing fluid in the quiescent state at normal fluid temperatures and pressures (standard conditions) Changes in the heat-extracting potential of the fluid brought about by nonstandard conditions of agitation, temperature, or pressure Product thickness and internal conditions of the part that affect heat flow to the surface Surface and other external conditions that affect the removal of heat Many different methods and quench media have been used in quenching aluminum alloys, depending on the alloy and product thickness and configuration. Hot or cold water and polyalkylene glycol (PAG) quenchants are used almost exclusively in the quenching of aluminum alloys. Water is the fluid most frequently employed for quenching aluminum, but there are a variety of quenchants and methods that have been used (Ref 1): Cold water immersion Hot water immersion Boiling water Water spray Quenching of Aluminum Alloys / 149 Quench Sensitivity of Alloys Polyalkylene glycol solutions Air blast Still air Liquid nitrogen Fast quenching oils Brine solutions Hot or cold water and PAG quenchants are used almost exclusively in the quenching of aluminum alloys, and these quenchants are discussed in separate sections of this article. Water is the fluid most frequently employed for quenching aluminum, and highly agitated cold water is an excellent quenchant in terms of obtaining high cooling rates. However, quenching in cold water can produce large differences in temperature between thick and thin sections, resulting in localized plastic flow and distortion observed after quenching or during machining. Usually, distortion is controlled in aluminum parts by adding polymers to water quenchants to reduce the convective or film coefficient between the part and the water, as discussed further in this article. Fig. 1 Effect of temperature on the extent of supersaturation and diffusion rate, which drive the rate of precipitation amounts of precipitation follow a C-shaped pattern (Fig.1). Using isothermal quenching techniques, Fink and Willey pioneered the attempts to describe the effects of quench rates with the use of C-curves (Ref 2). Using isothermal quenching techniques, they developed C-curves for strength of 7075-T6 and corrosion behavior of 2024-T4. The C-curves were plots of the time required at different temperatures to precipitate a sufficient amount of solute to either reduce strength (Fig. 2a), cause a change in the corrosion behavior (Fig. 2b), or relate to other properties, such as electrical conductivity, fracture toughness, or isothermal quench conditions. The nose of the C-curves identifies the region of highest precipitation rates, which Fink and Willey called the critical temperature range. For alloy 7075 (Fig. 2a), this range was determined to be 400 to 290 C (750 to 550 F). Investigators have used isothermal timetemperature-transformation diagrams to compare the quench sensitivity of aluminum alloys. Figures 3 and 4 are examples of isothermal transformation diagrams that compare the quench sensitivity of various aluminum alloys. The critical temperature range is roughly in the same range of 400 to 290 C (750 to 550 F), which is the approximate critical temperature range typical of many age-hardenable aluminum alloys. Some sources quote this range (or a slightly different range) as the most critical range for quenching of many aluminum alloys, but the range can be alloy-dependent. Quench sensitivity of aluminum alloys also has been evaluated from the effects of average quenching rates through the critical range from 400 to 290 C (750 to 550 F). In Fig. 5(a), for example, the effects of quenching on the yield strength of five alloys are compared in terms of average quenching rates through the critical range from 400 to 290 C (750 to 550 F). For alloys relatively high in sensitivity to quenching rate, such as 7075, rates of approximately 300 C/s (540 F/s) or higher are The most important metallurgical factor affecting the properties of age-hardenable aluminum alloys is solute loss, referring to the solutes that are chemically bonded with other elements and thus unavailable for precipitation hardening. There are several factors that can lead to solute loss and loss of properties. The rate of cooling in the part during quenching is one key factor, but solute loss also is affected by metallurgical factors such as the type of dispersoids in an alloy, solvus temperatures, and casting homogenization temperatures (see the article “Metallurgy of Heat Treatable Aluminum Alloys” in this Volume). During quenching, the objective is to prevent the precipitation of solutes during the cooling of high-temperature solid solution. If appreciable precipitation during cooling is to be avoided, two requirements must be satisfied. First, the time required for transfer of the load from the furnace to the quenching medium must be short enough to preclude slow precooling into the temperature range where very rapid precipitation takes place. The second requirement for avoidance of appreciable precipitation during quenching is that the volume, heatabsorption capacity, and rate of flow of the quenching medium be such that little or no precipitation occurs during cooling. Any interruption of the quench that may allow reheating into a temperature range where rapid precipitation can occur must be prohibited. The rate of precipitation, as a function of temperature, depends on two factors: the degree of supersaturation and the rate of diffusion (Fig. 1). Diffusion rates increase at higher temperatures, but the nucleation rate of precipitates is low because the degree of supersaturation is low at higher temperatures. Conversely, the precipitation rate also is low at low temperatures, where the degree of supersaturation is high but with low rates of diffusion. At intermediate temperatures, the precipitation rate is highest. Consequently, times to produce equal 900 900 800 800 600 100% 98% 80% 90% 300 500 400 200 300 200 10–1 Temperature, °F 700 Temperature, °C Temperature, °F 400 10 102 Fig. 2 300 500 200 300 200 10–1 103 Time, s (a) 600 400 400 Tensile strength Yield strength 1 Type of corrosion attack Pitting Pitting plus slight intergranular Pitting plus intergranular intergranular 700 Temperature, °C (b) 1 10 102 103 Time, s Time-temperature C-curve indicating the effect of time and temperature of an interrupted quench on (a) strength of alloy 7075 as a percentage of maximum from an uninterrupted quench and (b) type of corrosion attack in 2024-T4 sheet. Source: Ref 2 150 / Heat Treating of Aluminum and Its Alloys 500 700 800 930 7075 600 T6 temper 500 400 750 T6 temper Aged at 173 °C (340 °F) for 8 h 300 570 200 390 Temperature, °F 600 Temperature, °C 7050 Temperature, K Temperature, °F 700 500 400 300 .1 1 10 (a) Fig. 3 1 102 10 (b) 103 104 Critical time, s 1110 500 930 400 750 A B C D 800 425 700 370 6205 6063 600 315 300 570 200 390 500 260 212 400 20 A: 7075 B: 2017 C: 6061 D: 6063 100 32 0 1 (a) Fig. 4 6061 Temperature, °C 600 Temperature, °F Time-temperature C-curve indicating time and temperature of an interrupted quench on (a) yield strength of alloys 7075 and 7050 and (b) 99.5% maximum yield strength of 6351-T6 extrusion. Source: Ref 3–5 Temperature, °F Temperature, °C 100 Time, s 102 10 Time, s 103 1 (b) 0 102 103 Time of isothermal hold, s Examples of isothermal C-curves in comparing quench sensitivity of various aluminum alloys. (a) 95% of maximum tensile stress for various alloys. (b) 90% of maximum yield strength of 6xxx alloys in T6 temper. Source: Ref 6, 7 required to obtain near-maximum strength after precipitation heat treatment. The other alloys in Fig. 5(a) maintain their strengths at cooling rates as low as approximately 100 C/s (180 F/s). Figure 6 provides a similar comparison of agehardened tensile strength from different quench rates, including the effect of quench rate on stress corrosion of 2024. The effects of quench rates on tensile strength do not necessarily serve as the criterion to determine quench sensitivity. For example, the detrimental effects on resistance to corrosion of 2024-T4 occur at quenching rates considerably higher than those that mark the initial decrease in tensile strength (Fig. 7). Consequently, tensile properties do not serve as a criterion to determine whether quenching was sufficiently rapid to provide optimum corrosion resistance. However, in the case of 7075, the most rapid decrease in tensile properties occurs at cooling rates somewhat higher than those that have the greatest effects on corrosion (Fig. 7). To avoid completely the intergranular type of attack, rates in excess of approximately 165 C/s (300 F/s) for 7075-T6 and 555 C/s (1000 F/s) for 2024-T4 are needed. Such rates are not attainable with thick sections. Therefore, when thick-section parts are required to endure service conditions conducive to stress corrosion, artificially aged tempers of 2xxxseries alloy, in which precipitation is general within the grains, and the T73 special stresscorrosion-resistant temper of 7075 are preferred (Ref 11). Average quench rates through a critical temperature range can provide reasonable comparison of property in different alloys under similar product-process conditions (Fig. 8) and reasonable property predictions if cooling rates are fairly uniform. However, average quench rates do not reflect actual conditions under continuous cooling in a typical quench or with parts of variable section thickness. It is not necessarily sufficient just to ensure that the cooling curve misses the nose of the C-curve, because precipitation occurs throughout the cooling cycling—even when cooling curves involve holding times either above or below the critical temperature range. Under conditions of variable cooling rates over the entire quenching cycle, a more quantitative procedure, known as quenchfactor analysis, uses information from the entire C-curve to predict how a variable quench curve affects properties. See the section “QuenchFactor Analysis” in this article for more information. Quench-factor analysis is useful in evaluating the property effects when cooling rates are nonuniform, such as when cooling curves involve holding times either above or below the critical temperature range. Another useful method in evaluating quench sensitivity of alloys is the development of continuous cooling precipitation diagrams (see the article Quenching of Aluminum Alloys / 151 50 °C/s 100 Stress, ksi 80 Tensile strength 60 Yield strength 40 20 7178-T6 7075-T6 0 (a) 2 10 103 1 10 Quenching rate between 750 and 550 °F, °F/s (a) 500 °C/s 90 Strengths, ksi, or % loss by corrosion 120 80 70 Tensile strength 60 50 40 Yield strength 30 Stress corrosion 20 10 10 102 103 Average quenching rate, °F/s 1 (b) 102 Fig. 6 (b) Fig. 5 Quench sensitivity of various aluminum alloys as a function of average quench rates in the critical temperature range between 400 and 290 C (750 and 550 F). (a) Yield strength after aging of five wrought alloys. (b) Tensile strength after aging of eight wrought alloys Type Corrosion: I = intergranular, P = pitting, SI = slight intergranular 2024 I P+I P 7075 I P I or I+P Por P+SI Max depth, mils Attack in NaCl-H2O2 Quench sensitivity of various aluminum alloys as a function of average quench rates in the critical temperature range between 400 and 290 C (750 and 550 F). (a) Tensile and yield strengths of 7178-T6 and 7075-T6. (b) Tensile strength and corrosion properties of 2024-T4 sheet. Corrosion specimens were stressed to 75% of yield strength and exposed 48 h by alternate immersion in salt-peroxide solution. Corrosion losses are based on tensile strength. Source: Ref 8, 9 10 5 2024 7075 0 50 °C/s 500 °C/s Tensile strength, ksi 100 “Quench Sensitivity of Aluminum Alloys” in this Volume). More details on the effects of quench rates also are given in the article “Metallurgy of Heat Treatable Aluminum Alloys” in this Volume. 7075 80 2024 60 80 Quench Mechanisms 2024 Stressed to 75% of yield strength 60 Loss in tensile strength, % There are normally three distinct stages of quenching when a hot component is immersed in a liquid quenchant (Fig. 9, Ref 13): Vapor stage: Stage A or vapor blanket stage Boiling stage: Stage B or nucleate boiling Fig. 7 Unstressed 7075 20 0 10 stage Convection stage: Stage C These three general standard stages of quench mechanisms have very distinct effects on cooling rates (Fig. 9), although the different mechanisms may not be completely uniform over the surface of a given quenched part, depending on details of the quench method and the workpiece. For immersion quenching, quench rate commonly is defined conservatively as the rate of cooling when parts are fully immersed in the quenchant. Other nonstandard factors that can affect quench rate include time delay prior to full immersion and the degree of fluid agitation, which reduces the extent of 40 102 103 Average cooling rate (from 750 to 550 °F), °F/s 104 Effects of quenching rate on tensile properties and resistance to corrosion of 2024-T4 and 7075-T6. Source: Ref 10 vapor-phase formation, thus promoting more rapid onset of nucleate boiling (B stage) for a rapid cooling rate (Fig. 9). The first stage of the quench process is the vapor stage, which is encountered when the hot surface of the heated component first comes in contact with the liquid quenchant. The component becomes surrounded with a blanket of vapor. In this stage, heat transfer is very slow and occurs primarily by radiation through the vapor blanket. Some conduction also occurs through the vapor phase. This blanket is very stable, and its removal can only be enhanced by agitation or speed-improving additives. This stage is responsible for many of the surface soft spots encountered in quenching. 152 / Heat Treating of Aluminum and Its Alloys 110 7050 forgings Cold ST 475 °C (890°F) 4 h, quenched, water aged 120 °C (250 °F) 24 h quench 150 °F water quench Stress, ksi 90 Boiling water quench 600 80 500 70 Stress, MPa 100 7050 60 7075 7175 50 400 Fty L 7049 1 10 102 Average colling rate, °F/s 103 Fig. 8 Effect of average cooling rate after solution treatment on maximum yield sterngth of various aluminum alloys after aging. Source: Ref 12 High-pressure sprays and strong agitation eliminate this stage. If they are allowed to persist, undesirable microconstituents can form. The second stage encountered in quenching is the boiling stage. This is where the vapor stage starts to collapse and all liquid in contact with the component surface erupts into boiling bubbles. This is the fastest stage of quenching. The high heat-extraction rates are due to carrying away heat from the hot surface and transferring it further into the liquid quenchant, which allows cooled liquid to replace it at the surface. In many quenchants, additives have been added to enhance the maximum cooling rates obtained by a given fluid. The boiling stage stops when the temperature of the component surface reaches a temperature below the boiling point of the liquid. For many distortion-prone components, elevatedtemperature or polymer quenchants are used if the medium is fast enough to maintain supersaturated solid solution in quenched aluminum. The final stage of quenching is the convection stage. This occurs when the component has reached a point below that of the quenchant boiling temperature. Heat is removed by convection and is controlled by the quenchant specific heat and thermal conductivity and the temperature differential between the component temperature and that of the quenchant. The cooling rate during the convection stage is usually the slowest of the three stages. Typically, it is this stage where most distortion occurs. Standards Tests for Cooling Curves. The most useful way of accurately describing the complex mechanism of quenching is to develop a cooling curve for the quenching medium under controlled conditions. Cooling curves are developed by quenching, from an elevated temperature, a test probe in a sample of the quenching medium. Sometimes an austenitic stainless steel specimen is used to avoid scaling or the necessity for a protective atmosphere. A high-speed recorder or digital data-acquisition unit is used for plotting Fig. 9 Cooling stages and mechanisms during immersion quenching. Source: Ref 13 temperature changes, as measured by one or more thermocouples embedded in the probe. The resulting time-temperature curve indicates the heat-transfer characteristics of the quenching fluid. In general practice, the use of an Inconel probe such as that described by ASTM D6200 is used. This precludes the issues of oxidation and latent heats of transformation. This type of probe is the most commonly used probe. However, other probes manufactured from silver or other materials can be used. The probe material should always be specified. It is generally not possible to compare probes of different materials and tested to different methods. A comparison of the different cooling curve methods and probe designs is shown in Table 1. Quench Severity and Cooling Rates Several material and quenchant characteristics influence the rate of heat removal from the part being quenched. An infinite quench is one that instantly decreases the skin of the part to the bath temperature. In practice, quenchants never provide an idealized infinite quench, but with a very rapid rate of cooling at the surface, the rate of cooling in the part is then a function only of the diffusivity of the metal, that is, its ability to diffuse heat from the interior to the surface. The rate of cooling in production operations, which is not always easily measured, depends on several factors: Type of quench fluid (water, air, polymer, liquid nitrogen) Quench method (immersion, spray, still air, air blast) Thickness, configuration, and surface condi- tion of the parts Internal conditions of the workpiece that affect the supply of heat to the surface Surface and other external conditions that affect the removal of heat Heat-extracting potential of the quenching fluid in the quiescent state at normal fluid temperatures and pressures (measured under standard conditions with various probes, e.g., see the section “Standards Tests for Cooling Curves” in this article) Changes in the heat-extracting potential of the fluid brought about by nonstandard conditions of agitation, temperature, or pressure The severity of the quench at the surface depends, in practice, on the vapor blanket formation, boiling characteristics, and the velocity, temperature, specific heat, heat of vaporization, conductivity, density, viscosity, and wetting characteristics of the quenching fluid. Practically, the cooling rates are controlled by the fluid selected, the type and amount of any polymer put in water-based quenchants, and the bath temperature and velocity. Quench rate also depends on several product factors, such as part thickness and surface conditions. As the thickness is increased, a slower cooling rate occurs at the interior and midplane of the part. If the quench rate is slow enough, or the product thickness is large, then significant precipitation can occur. For example, cooling rates determined experimentally for 1.6 mm to 20 cm (0.06 to 8 in.) thick sections that were quenched by immersion in water at five different temperatures and by cooling in still air are shown in Fig. 10. The dashed line at the extreme right in Fig. 10 delineates the maximum theoretical cooling rate at the midplane of the plate. This assumes an infinite cooling rate and a thermal diffusivity of 1400 cm2/s. This cooling rate assumes that the surface of the plate has instantaneously changed to the temperature of the quenchant. This is often used as a limiting case Quenching of Aluminum Alloys / 153 Table 1 Cooling curve test methods Method JIS K2242 Z8 E 45003 ASTM D6200 International Inconel 600 12.5 60 (0.5 2.4) Various 115 ± 5 (4.5 ± 0.2) diam 2000 (68) 40 ± 2 (105 ± 4) 850 ± 5 (1560 ± 9) France 99.999% Ag 16 48 (0.6 1.9) Various 138 diam 99 high (5.4 3.9) 800 (27) 50 ± 2 (120 ± 4) 800 ± 5 (1470 ± 9) Japan 99.999% Ag 10 30 (0.4 1.2) Dioctyl phthalate 300 mL (10 oz) beaker 250 (8.5) 80, 120, 160 (175, 250, 320) 810 ± 5 (1490 ± 9) China 99.999% Ag 10 30 (0.4 1.2) Dioctyl phthalate 300 mL (10 oz) beaker 250 (8.5) 80 ± 2 (175 ± 4) 810 ± 5 (1490 ± 9) United States Inconel 600 12.5 60 (0.5 2.4) Various 115 ± 5 (4.5 ± 0.2) diam 2000 (68) 40 ± 2 (105 ± 4) 850 ± 5 (1560 ± 9) Average colling rate at 400–290 °C, °C/s 1 10 102 10–1 103 25 1 Thickness, in. Thickness, mm 10 Computed maximum (assumes instantaneous cooling of surface from 875–210 °F or 470–100 °C) 75 7.5 Air cool 212 °F 180 °F (100 °C) 150 °F (80 °C) 200 °F (65 °C) (95 °C) Immersion in water at indicated temperature 0.75 1 10 102 Average colling rate at 750–550 °F, °F/s 103 Q ¼ h AðTS T1 Þ (Eq 1) where Q is the amount of heat transferred, A is the surface area of the part in contact with the fluid, TS is the surface temperature, T1 is the fluid temperature away from the surface, and h is the interfacial or film coefficient. If this equation is rearranged, h, the film coefficient, can be defined in terms of the part area, difference in temperature between the part and the quenchant, and the heat being transferred. The rate of heat transfer at the surface is thus: 200 6 150 Square 4 100 Round 2 50 2 4 6 8 10 Section size, in. 12 14 Experimentally determined correlation between the average cooling rates of 400 to 290 C/s (750 to 550 F/s) of rod and square bars to plates. Rates were measured at the centers of sections. Source: Ref 14 75 °F (25 °C) 10–2 104 Effects of thickness and quenching medium on average cooling rates at midplane of aluminum alloy sheet and plate quenched from solution temperatures. The dashed line delineates the maximum cooling rates theoretically obtainable at the midplane of plate, assuming an infinite heat-transfer coefficient and a diffusivity factor 2 of 1400 cm /s. Source: Ref 14 dQ ¼ hðTS T1 Þ dt S Section size, mm 100 150 200 250 300 350 250 Fig. 11 Fig. 10 for quenching. No rates higher than this can be achieved, although rates approaching the theoretical limit have been observed with impinging spray quenching. Experimentally determined relationships between the thickness of plates and either the diameter of rounds or the dimensions of square bars having equal cooling rates are shown in Fig. 11. Quench Severity at the Surface. Mathematically, heat transfer at the surface of the part can be described using Newton’s law of cooling: 50 8 0 0 10–1 2.5 0.25 10–1 10 Thickness of plate having equal cooling rate, mm 250 AFNOR NFT 6077B Thickness of plate having equal cooling rate, in. Country Probe alloy Probe dimensions, mm (in.) Reference fluid Vessel dimensions, mm (in.) Oil volume, mL (oz) Oil temperature, C ( F) Probe temperature, C ( F) ISO 9950 (Eq 2) such that TS is time-dependent. The greatest conceivable quench severity would be when the surface is cooled instantaneously to that of the quenchant. A less severe quench leads to less rapid temperature reduction. An analytical determination of the interface coefficient, h, requires that the properties of the fluid moving past the part be examined. The properties of the quenchant, including boiling temperature, viscosity, density, thermal conductivity, and specific heat, combine to make the quenchant an important, if not the most important, variable affecting quench severity. Increases in quenchant velocity generally increase the quench severity. Increasing the bath temperature puts the quenchant nearer its boiling point, decreases the temperature difference between the part and bath, and decreases the quench severity. Quench severity and cooling rates also are very sensitive to the surface condition of the parts. Lowest rates are observed with products having freshly machined or bright-etched, clean surfaces, or products that have been coated with materials that decrease heat transfer. The presence of oxide films or stains increases cooling rates. Further marked changes can be effected through the application of nonreflective coatings, which also accelerate heating (Fig. 12). Surface roughness exerts a similar effect; this appears to be related to vapor film stability. In addition, the manner in which parts enter the quenching medium can significantly alter the relative cooling rates at various points, thereby affecting mechanical properties and residual stresses established during quenching. In batch heat treating operations, placement and spacing of parts on the racks can be a major factor in determining the quenching rates. In immersion quenching, adequate volumes of the quenching medium must be provided to prevent an excessive temperature rise in the medium. When jet agitation is used to induce water flow between parts, jets should not impinge directly and cause rapid localized cooling Heat-Transfer and Cooling Rates within the Part. Cooling rates in the part depend on the size and configuration of the part. Similarly, quenching complex shapes (such as engineered castings, die forgings, or extruded shapes whose wall thicknesses differ widely) poses special problems if distortion and stresses are to be minimized. If all workpieces were symmetrical and alike in shape (no odd 154 / Heat Treating of Aluminum and Its Alloys of the part, and dT/dx is the thermal gradient in the part. To obtain the temperature distribution and gradients within a part over time requires the use of Fourier’s second law of heat conduction, which, in simplified form for one dimension, is: 2 dT d T ¼a dt dx2 (Eq 8) where a is the thermal diffusivity of the metal part, which is related to density (r), specific heat (Cp), and thermal conductivity as k = a r Cp. Solution of the differential equations for Fourier’s second law under appropriate boundary conditions (e.g., surface temperature, part shape, part size) requires numerical integration of the differential equations for heat transfer. In the case of heat transfer from the interior of a bar, while neglecting axial flow, the relation is: d2 T 1 dT 1 dT þ ¼ dr 2 r dr dt (Eq 9) where r is the bar radius, a is the thermal diffusivity, and dT/dr is the thermal gradient. The thermal conductivity (k) of the part can be related to the film coefficient (h) of the bath using Biot’s number (Bi) in the equation: Biot’s numberðBi Þ ¼ hX=k (Eq 10) where X is the characteristic length of the part. A similar ratio, more widely used in quenching, is the Grossman number, defined by the equation: Fig. 12 Effect of surface conditions on the midplane cooling of a 13 mm (0.5 in.) thick plate of 7075 from quenching in (a) 20 C (70 F) water and (b) boiling water configurations) and were of the same size and/ or weight, obtaining the desired properties would be simple. In practice, however, such conditions rarely exist, and the cooling rate depends on the internal conditions of the workpiece that affect the supply of heat to the surface. Because heat transfer during quenching basically is limited by resistance at the surface in contact with the quenching medium, the average rate of cooling can be estimated as a function of the ratio of surface area to volume. This ratio may vary considerably, depending on the shape of the product. For sheet and plate, as well as other products of similar shape, average cooling rates (through the critical temperature range measured at a center or midplane location) vary with thickness in a relatively simple manner. The relation can be approximated by: logðR1 Þ ¼ logðR2 Þ C logðtÞ More generally, the flux of heat and temperature gradient within a part are related by Fourier’s equation: dQ dT ¼k dt dx (Eq 4) where k is the thermal conductivity of steel. At the surface, the temperature gradient is: dQ dT ¼k dt dx S (Eq 5) So that at the surface: dT h ¼ ðTS T1 Þ dx S k (Eq 6) The actual heat flow over a surface area, A, from the interior to the surface of a part being quenched can be described with Fourier’s equation: (Eq 3) where R1 is the average cooling rate at thickness t, R2 is the average cooling rate at 1 cm (0.4 in.) thickness, and C is a constant. Q ¼ k A dT=dx (Eq 7) where Q is the amount of heat transferred, k is thermal conductivity of the alloy, A is the area H ¼ h=2k (Eq 11) The Grossman number has been reported to equal approximately 1 for 25 mm (1 in.) sections quenched in still water. As an example in the following sections (from Ref 15), a finite-difference heat-transfer program was used to solve Eq 9 for various experimental conditions with cylindrical (bar) test probes. Grossman numbers (H) and the film coefficient (h) provide useful information about the rate of heat removal from the surface of a part. From experimental data of cooling curves obtained from quenching solution-treated 7075 probes under controlled conditions, Grossman numbers (H) and film (heat-transfer) coefficients were calculated for water quenches under a range of velocity and temperature conditions and with additions of polymers under selected conditions (Table 2). The 7075 aluminum probe had a length of 280 mm (11 in.) and a diameter of 75 mm (3 in.), with thermocouples inserted in the centerline and 6.35 mm (0.25 in.) from the surface. Probes were solution treated at 465 C (870 F), and experimental data of the cooling curves are given in Fig. 13. The temperature difference between the two thermocouples is also plotted. With the thermal conductivity of 7075 aluminum at approximately 1.70 W/cm K (1150 Quenching of Aluminum Alloys / 155 Btu in./ft2 h F), Grossman numbers (H) and film (or heat-transfer) coefficients were calculated. Unagitated water at 25 C (75 F) has a Grossman number of approximately 1, as reported by Grossman, and a film coefficient of approximately 3.55 W/cm2 K (2460 Btu in./ft2 h F). The film coefficient increased to 4.78 and 5.14 W/cm2 K (3105 and 3565 Btu in./ft2 h F) at velocities of 0.25 and 0.50 m/s (50 and 100 ft/min), respectively. These high film coefficient values can therefore cause cold water quenching to create high thermal Table 2 Grossmann numbers and heat-transfer coefficients (C) of quenchant-to-part films Quenchant F m/s ft/min Grossmann number (H = C/2k) 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0.00 0.25 0.50 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 0 50 100 1.07 1.35 1.55 0.99 1.21 1.48 1.10 1.29 1.60 0.86 1.09 1.33 0.21 0.57 0.79 0.11 0.21 0.27 0.06 0.08 0.09 0.04 0.04 0.04 0.19 0.21 0.23 0.44 0.40 0.42 Temperature Type C Water 27 80 Water 38 100 Water 49 120 Water 60 140 Water 71 160 Water 82 180 Water 93 200 Water 100 212 Polyalkylene glycol (UCON A)(a) 30 85 Polyvinyl pyrrolidone (PVP 90)(a) 30 85 Velocity Effective heat-transfer coefficient (C) W/cm2 K Btu/ft2 h F 3.55 4.78 5.14 3.28 4.01 4.91 3.65 4.29 5.31 2.85 3.62 4.41 0.70 1.89 2.62 0.36 0.69 0.89 0.20 0.27 0.30 0.13 0.13 0.13 0.63 0.70 0.77 1.49 1.34 1.41 2460 3105 3565 2275 2785 3400 2530 2970 3680 1980 2510 3060 485 1310 1815 255 485 620 138 184 207 92 92 92 429 475 529 1012 912 966 (a) Polymer quenchants with concentrations of 25%. k is equal to the thermal conductivity of the aluminum alloy (7075). Source: Ref 15 550 1000 550 1000 500 900 500 900 450 800 800 250 200 400 Temperature, °C 300 ΔT in 60 °C (140 °F) water 700 Temperature, °F Temperature, °C 400 350 ΔT in 32 °C (90 °F) water 600 500 400 Cooling curves in 60 °C (140 °F) water 350 300 250 200 500 400 300 200 100 200 50 100 50 100 0 0 0 0 300 100 0 (a) 20 40 60 80 100 ΔT in 32 °C (90 °F) water at 0.25 m/s (50 ft/min) 600 150 150 ΔT in 25% PQ90, 0.25 m/s (50 ft/min) 32 °C (90 °F) 700 Temperature, °F 450 gradients from surface to center of a part and high temperature differences between thick and thin sections. The film (heat-transfer) coefficients generally decreased with increasing water temperature until, at temperatures of 90 to 100 C (195 to 212 F), the film coefficients were in the range of 0.13 to 0.30 W/cm2 K (92 to 207 Btu in./ft2 h F). Table 2 provides similar data on two polymer solutions under selected conditions. The 25% solution of polyalkalene glycol (UCON A) produced Grossman numbers of 0.19 to 0.23 and film coefficients of 0.63 to 0.77 W/cm2 K (429 to 529 Btu in./ft2 h F) when the bath was operated at 30 C (85 F) and with velocities from 0 to 0.5 m/s (0 to 100 ft/min). Polyvinyl pyrrolidone (PVP) 90 produced Grossman numbers of 0.40 to 0.44 and film coefficients of 1.34 to 1.49 W/cm2 K (912 to 1012 Btu in./ft2 h F) under these conditions. The higher film coefficient of PVP 90 suggests that it can be used to quench heavier sectioned parts. If the Grossman number, H, or the effective interface heat-transfer coefficient, h, between the part and the quenchant is established, the quench factor in commercial shapes can be calculated using finite-element or finite-difference heat-transfer programs. The results of calculations on sheets and plates made using constant film (or heat-transfer) coefficients are illustrated in Fig. 14. These figures illustrate the interrelationship between aluminum sheet or plate thickness, film coefficient, and quench factor. The calculations for these graphs were made using film coefficients indicated at the end of each diagonal line. The diagonal lines represent lines of constant film coefficient. The important feature of these figures is that (with data on film coefficient such as that Cooling curves in 25% PQ90, 32 °C (90 °F) Cooling curves in 32 °C (90 °F) water 0 20 40 60 80 100 150 200 250 Time, s Time, s 0 50 100 ΔT, °F 0 (b) Fig. 13 20 40 60 80 100 120 140 ΔT, °C Cooling curves and temperature difference across a 76.2 mm (3 in.) diameter 7075 alloy probe with quenchant flow of 0.25 m/s (50 ft/min). (a) Water quenchant at 60 and 32 C (140 and 90 F). (b) Water quenchant and 25% PQ90 quenchant at 32 C (90 F). Source: Ref 15 156 / Heat Treating of Aluminum and Its Alloys presented in Table 2) estimates can be made about the ability of specific quenchants and operating conditions to provide cooling rates sufficiently high to meet minimum mechanical properties in parts of various thicknesses. (See the section “Quench-Factor Analysis” in this article for more details.) specific heat of vaporization and high specific heat capacity. The thermal conductivity is very small compared to most metals. Therefore, water is used wherever it is practical, that is, where the drastic quench afforded by water does not result in excessive distortion or warping of the workpiece. Other advantages of water include: Water as a Quenchant C 20 15 10 5 1.25 (a) 4 .14 =0 16 0.2 C = 0.288 C= 32 C = 0.4 20 C = 0.7 C = 1.44 2.5 3.75 5 6.25 7.5 Sheet thickness, mm = 1. 15 C= C= 0.7 2 0.4 3 .216 44 1. 16 8 C 2.8 = C 4.32 = 0 C .2 =7 C 25 = 20 15 10 2. 5 0 0 12.5 25 37.5 50 Plate thickness, mm (b) 62.5 75 Plots of quench factors derived from finite-element analysis with given (a) sheet and (b) plate product sizes and film (heat-transfer) coefficients (C). Heat-transfer coefficients between the quenchant and part are expressed in W/cm2 K. Source: Ref 15 1000 1800 100 Cooling rate, °F/s 150 1800 1600 1400 800 Temperature, °C 1200 800 1400 1200 600 1000 800 400 Water temperature: 40 °C 50 °C 60 °C 70 °C 80 °C 90 °C 600 400 200 200 0 0 (a) Fig. 15 10 20 30 Time, s 40 50 250 1600 1000 200 200 1000 600 400 50 Temperature, °F Water temperature: 40 °C 50 °C 60 °C 70 °C 80 °C 90 °C 800 Temperature, °C C=0 30 0 60 (b) 0 20 40 60 80 100 120 140 Temperature, °F 25 = 35 = C 40 3 C 8 10 0. 45 2.5 C 30 50 Sheet thickness, in. 1 1.5 2 0.5 0 C = 0.072 2 07 0. C 0.0 35 = 36 40 Fig. 14 0.3 •K W/ 45 0 0 0.25 One disadvantage of plain water as a quenchant is that its rapid cooling rate persists throughout the lower temperature range, contributing to large thermal stress and distortion Quench factor for 99.5% of attainable yield strength for alloy 7075-T73 Sheet thickness, in. 0.1 0.15 0.2 0.05 cm 2 0 50 C= Quench factor for 99.5% of attainable yield strength for alloy 7075-T73 Water is the fluid most frequently employed for quenching aluminum, and it is widely used for quenching all aluminum alloys. Water quenching is done either by total immersion or by spray. As a quenching medium, plain water approaches the maximum cooling rate attainable in a liquid. Water has a high quenching power (heat-transfer coefficient) due to the high Lack of flammability Low cost No health hazards Easy scale removal by filtration No environmental hazards associated with water in quenched parts. Another disadvantage of using plain water is that its vapor blanket stage may be prolonged. This prolongation, which varies with the degree to which the complexity of the part being quenched encourages vapor entrapment and with the temperature of the quench water, results in uneven hardness, unfavorable distribution of stress, and distortion. To obtain reproducible results by water quenching, the temperature, agitation, and contamination must be controlled. Water temperature is the largest primary variable controlling the cooling rate. Surface cooling power of water decreases rapidly as water temperature increases Fig. 15. As the temperature of water is raised, the stability of the vapor phase increases, and the onset of nucleate boiling in a stagnant fluid is suppressed. Agitation helps dispel the prolonged vapor phase and increases the cooling rate (Fig. 16). Agitation is especially important in water quenching because it provides for more uniform heat transfer around parts. If the water is not well agitated, then highly variable heat transfer can occur. This is especially true of nonsymmetrical parts, where large differences in heat transfer can result in distortion or cracking. Very large differences in heat transfer also can occur when the vapor phase persists in holes, cavities, and on surfaces with adjacent parts. Water Contamination. An early study using cooling curves (Ref 16) showed that quenching into still water caused rapid heat transfer. This study showed that heat transfer at the surface of the part was very turbulent at the metal/water interface. This study also showed that there was a marked difference between hard water and distilled water. Distilled water showed an extensive vapor blanket that extended to very low temperatures (Fig. 17). If a water quenchant contains foreign substances, such as emulsions, dissolved salts, 600 400 200 160 Cooling rate, °C/s Effect of bath temperature on heat removal in an ASTM D6200 probe. (a) Cooling curves. (b) Cooling rate curves. Quenchant is water having 0.25 m/s (50 ft/min) velocity. Quenching of Aluminum Alloys / 157 The first groups of contaminants are those that are poorly soluble in water. Solids such as soot and liquids that contain soaps, fats, and oils form suspensions or emulsions that are prone to surface reactions that promote vaporphase stability. This results in an increased duration of the vapor phase, with a lower temperature of nucleate boiling initiation. Oils, soaps, and fats are the most damaging. As a result, this group increases nonuniform heat transfer. This nonuniformity manifests itself as spotty hardness or increased distortion. Dissolved gases behave in a similar fashion, with increased dissolved gases coming out of solution during the quenching process. This is one reason why compressed air is not recommended for agitating water quenchants. Salts, acids, and alkalis readily dissolve in water. These act to reduce the stability of the vapor phase during quenching. If the concentration is high, then the vapor phase will not form at all. This type of contamination can be taken advantage of to create quenchants with very fast quench rates. Brine Quenching. Addition of a low percentage of salt (up to 10%) increases the speed of quenching over that of cold water. The saltwater solution tends to break up the vapor phases, thus increasing cooling power. It is only rarely used when water is not sufficient in obtaining properties in thicker sections. Cooling curves and cooling rate curves for a 25 mm (1 in.) diameter stainless steel probe quenched in 55 C (130 F) water that is flowing at selected velocities from 0 to 0.75 m/s (0 to 150 ft/min) 1650 900 Temperature, °C 800 Hard water 20 °C (70 °F) 40 °C (105 °F) 60 °C (140 °F) Distilled water 20 °C (70 °F) 40 °C (105 °F) 60 °C (140 °F) 1470 700 1290 600 1110 500 930 400 750 300 570 200 390 100 212 Temperature, °F Fig. 16 32 0 0 10 20 30 40 50 60 Time, s Fig. 17 Comparison of hard and distilled water cooling curves at different temperatures or gases, the quenching characteristics can be drastically altered. Local ground water or tap water can have large differences in soluble gases, salts, or solids. This results in different localities having different quenching characteristics. As was noted previously, the most important factor of water quenching is the persistent vapor phase. Based on this, contaminants may be divided according to their effect on the vapor phase, that is, either increasing or decreasing the stability of the vapor phase. Immersion Water Quenching For most aluminum applications, either cold or hot water immersion quenching is performed. Maximum properties are obtained with quenching in cold water, while distortion control and residual stresses are minimized with quenching by hot water at 70 C (160 F) or more. In quenching some products, water at below 38 C (100 F) provides the required quench rate for optimum properties of the alloy being heat treated. In others, the water may be purposely heated to the boiling point to control distortion and residual stresses. Some normally accepted water quenching practices for common aluminum alloys are summarized in Table 3. Quench rates (defined as the cooling rates when the parts are fully immersed in the quenchant) depend primarily on water temperature and agitation. In addition, two other factors influence the extent of unintended precipitation (solute loss) after solution treatment: Quench delay: the total time from opening the furnace until the parts are submerged in the quenchant (or otherwise fully subjected to a quench) Immersion rate: the rate at which parts enter the quenchant (or quench system) The cooling rate depends primarily on water temperature and agitation (Ref 17). The cooling 158 / Heat Treating of Aluminum and Its Alloys rate of water quenching is independent of material properties such as thermal conductivity and specific heat. Water temperature is the largest primary variable controlling the cooling rate. Surface cooling power of water decreases rapidly as water temperature increases (Fig. 15). Hot water has a low cooling power because, as the boiling point is approached, the vapor phase becomes prolonged. Agitation is important, because it provides more uniform heat transfer around parts by disrupting the prolonged formation of vapor phase. As agitation is increased, the variation in hardness decreases and distortion decreases. Water at a temperature of 15 to 25 C (60 to 75 F) can provide uniform quenching speed and reproducible results. Quenching into water at less than 50 to 60 C (120 to 140 F) often produces nonuniform quenching. This nonuniformity manifests itself as spotty hardness, distortion, and cracking. This nonuniformity is caused by relatively unstable vapor blanket formation. Because of this difficulty, immersion quenching in cold water usually is restricted to the quenching of simple, symmetrical parts. Polyalkylene glycol quenchants also are used to provide a quench rate in between that of water and oil. By control of agitation, temperature, and concentration, quench rates similar to water can be achieved. Because rapid cooling rates are achieved with water at lower temperatures, water near room temperature (15 to 25 C, or 60 to 75 F) is used for many aluminum quenching operations. The temperature at the onset of quenching is usually in the range of 15 to 30 C (60 to 90 F). Most specifications limit the temperature of the water to below 30 C (90 F), with the maximum rise of no more than 5 C (10 F). This requirement governs the design of most quench tanks regarding the total volume in an immersion quench tank. (See the section “Quench Tank Systems” in this article for more details.) Effect of Water Temperature. When water quenchant temperature is increased, two things occur. First, the vapor phase becomes much more pronounced and stable. Second, the maximum cooling rate during nucleate boiling decreases. In addition, the temperature of maximum cooling also decreases as the temperature of the water is increased (Table 4). These effects also are illustrated in the cooling curves of Fig. 15. In general, as the temperature of water is raised, the stability of the vapor phase increases, and the onset of nucleate boiling in a stagnant fluid is suppressed. Table 5 summarizes the effects of water temperature on cooling rates for aluminum alloys of different thicknesses. When water temperature is raised above 70 C (160 F), the quenching rates are drastically reduced (Fig. 18a). This can result in a significant loss of strength when heat treating the more quench-sensitive 2xxx and 7xxx alloys, such as 7075, 7049, and 7178. For example, Fig. 19 illustrates the effect of quench rate on the strength of 7075 plate with a thickness of 13 mm (0.5 in.). Significant losses in water lowers strength even further but has the beneficial effect of lowering residual stresses below that of cold or hot water quench. The compromise is to reduce attainable strength in favor of a more dimensional stability. However, depending on the agitation within the tank, quench speeds in boiling water are sometimes strength of thicker products also can occur with a water temperature of 50 C (120 F). Because of the rapid change in quenching characteristics of water above 70 C (160 F), close control of agitation is required to ensure better control of quenching rates at various locations in the tank. Quenching in boiling Table 3 Water quenching practices for common aluminum alloys Cold water Form Alloy/temper 20–32 C (70–90 F) Sheet 2014 2024 2219 6061 7075 7049 7050 7175 2024 2219 6061 7075 7049 7050 7175 2014-T6 2014-T61 2024 2219 6061 7075 7049 7050 7175 C355 A356 A356 premium A357 premium A201 X X X X X X X X X X X X X X X X ... ... ... ... ... ... ... X ... ... X X X Plate Forgings Castings Hot water Boiling water 55–65 C (130–150 F) 60–70 C (140–160 F) 65–100 C (150–212 F) 95–100 C (202–212 F) ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... X ... ... X X X ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... X ... X X X X ... X ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... ... X ... ... ... ... ... ... ... ... ... ... ... ... Source: Ref 1 Table 4 Effect of water temperature on cooling rates Water temperature C 40 50 60 70 80 90 Maximum cooling rate F 105 120 140 160 175 195 C/s 153 137 115 99 79 48 Maximum cooling-rate temperature F/s 275 247 207 178 142 86 C 535 542 482 448 369 270 F 995 1008 900 838 696 518 Cooling rate at specified temperature, C/s ( F/s) 704 C (1299 F) 60 32 20 17 15 12 (108) (58) (36) (31) (27) (22) 343 C (649 F) 97 94 87 84 77 26 (175) (169) (157) (151) (139) (47) 232 C (450 F) 51 51 46 47 47 42 (92) (92) (83) (85) (85) (76) Source: Ref 18 Table 5 Effects of water temperature on cooling rates for aluminum alloys of different thicknesses Quenching rate for specified thickness, C/s ( F/s) Water quenchant C 32 55 70 88 100 Source: Ref 1 F 90 130 160 190 212 0.75 mm (0.030 in.) 5,600 1,700 500 140 50 (10,000) (3000) (900) (250) (90) 1.75 mm (0.070 in.) 3,300 560 170 60 17 (6000) (1000) (300) (105) (30) 25 mm (1.00 in.) 75 mm (3.00 in.) 120 (220) 90 (160) 35 (65) 4.5 (8) 2 (4) 16 (28) 12 (22) 8 (15) 1.7 (3) 1 (2) Quenching of Aluminum Alloys / 159 900 Table 6 Typical quench delay times for aluminum alloys of various thicknesses 400 200 700 600 300 500 400 75 300 200 0 130 4 200 160 8 (a) 12 16 Minimum thickness Temperature, °C Temperature, °F 800 mm Up to 0.41, inclusive Over 0.41 to 0.79, inclusive Over 0.79 to 2.29, inclusive Over 2.29 20 Time, s in. Up to 0.016, inclusive Over 0.016 to 0.031, inclusive Over 0.031 to 0.090, inclusive Over 0.090 Maximum time, s 5 7 10 15 Source: Ref 19 900 800 60% 600 300 500 400 200 40% 300 H2O 200 0 (b) Temperature, °C Temperature, °F 400 700 30% 12% 4 20% 8 12 16 20 Time, s Fig. 18 Comparison cooling rates for 12.5 mm (0.5 in.) thick wrought 7075 aluminum plate when (a) quenching with water at different temperatures and (b) quenching with different concentrations of AMS type I polymer quenchant uneven, resulting in uneven residual stresses. When quenched in boiling water, the part is actually being quenched in a continuous vapor pocket, especially during the early stages of the quench (Ref 1). The alternative is to consider PAG quenchants, which can achieve higher strength and lower residual stress compared to a boiling water quench. In practice, aluminum parts prone to distortion (especially castings and forgings) are quenched in either hot or boiling water. In the case of other wrought products, such as plates and extrusions, only the less quench-sensitive alloys (such as 6061) are quenched in boiling water. In some other specific applications, boiling water may be used to achieve a high level of dimensional stability. For example, alloy 2014 has a specific temper (2014-T61) of a boiling water quench for dimensional stability (Ref 1). There is relatively little impact of increasing water temperature on C-stage cooling rates. Because the objective of raising the water temperature is to decrease cracking and distortion, and because the tendency for cracking to occur is often proportional to C-stage cooling rates, these figures show that raising the water temperature is a relatively ineffective procedure. These data explain why alternative quenchants such as aqueous polymers are used. Another reason for the use of these quenchants as alternatives to hot water is that these media produce much more uniform wetting of the metal surface during the quenching. This is critically important if localized surface cracking and distortion are to be avoided. The interaction of temperature and agitation on the vapor phase is much stronger for water Fig. 19 Effect of water temperature on the strength of 7075 plate with a thickness of 13 mm (0.5 in.) than in other quenchants. This is why water quenching is generally not used for complex shapes or blind holes. These geometrical obstructions result in a nonuniform heat transfer around the surface of the part. In areas of persistent vapor phase, poor quenching and solute precipitation can occur. Quench Delay. Whether the transfer of parts from the furnace to the quench is performed manually or mechanically, it must be completed in less than the specified maximum time. The maximum allowable transfer time, or quench delay, varies with the temperature and velocity of the ambient air and the mass and emissivity of the parts. The maximum quench delays (Table 6) can be determined that will ensure complete immersion before the parts cool below 400 C (750 F). Aerospace Materials Specification (AMS) 2770 specifies maximum quench delays for high-strength alloys of 5, 7, 10, and 15 s for thickness ranges of up to 0.41 mm (0.016 in.), 0.41 to 0.79 mm (0.016 to 0.031 in.), 0.79 to 2.29 mm (0.031 to 0.090 in.), and over 2.29 mm (0.090 in.), respectively. Quench delay is conservatively defined as commencing “when the furnace door begins to open or the first corner of a load emerges from a salt bath” and ending “when the last corner of the load is immersed in the water quench tank.” Recommended maximum quench delay times are listed in Table 6. However, exceeding the maximum delay time is permitted if temperature measurements of the load prove that all parts are above 415 C (775 F) when quenched. The C-curves used in quench-factor analysis can also assist in determining a maximum allowable delay. It is relatively easy to control quench delay in day-to-day operations by using a stopwatch or, if necessary, by attaching thermocouples to parts. However, although the cooling rate between 400 and 260 C (750 and 500 F) is most critical and must be extremely high for many high-strength alloys, it cannot be directly measured in production operations. It is usual to rely on standardized practices, augmented by results of tension tests and tests of susceptibility to intergranular corrosion. Immersion Rate Control. Control of immersion rates is very important to minimize warpage when quenching sheet or thin-gage products. A faster immersion rate results in less distortion, and more modern furnaces are designed with controls to select immersion rates. Immersion rates are normally in the range of 0.15 to 3.0 m/s (0.5 to 10 ft/s) (Ref 1). Spray or Fog Quenching Spray techniques vary from the application of a heavy spray to that of a light mist or fog. Spray quenching is confined almost entirely to products having one long dimension, including extrusions, rolled structural shapes, sheet, and plate. Because spray quenching avoids the hazard of steam pocketing, it can result in a vigorous quench. However, it requires judicious design and application to avoid nonuniform cooling that would cause undesirable distortion. Spray quenching generally is accomplished by passing the product through a chamber containing a multiplicity of spray nozzles, arranged to provide thorough, complete coverage of all exposed surfaces. The entry sprays are most important. Unless the entire periphery of the metal is contacted uniformly, distortion and bowing occur. The nozzles for these sprays are selected and arranged to deliver a flat pattern; if sections of variable height are to be processed, the nozzles should be adjustable. Flooding-type or square-pattern nozzles usually follow the entry sprays. The spray chamber should be long enough to permit a rate of metal movement into the sprays that avoids excessive precooling of the rear of the charge before that part reaches the lead spray. The terminal sprays at the discharge end frequently are directed into the quench chamber to minimize carry-out of quench medium; they may be followed by airblast nozzles to reduce this possibility further. If possible, cooling performance should be verified by cooling curves in the production chamber. For spray quenching, the quench rate is controlled by the velocity of the water and by volume of water per unit area per unit time of impingement of the water on the workpiece. Spray quenching rates are much slower than even high-concentration polymer quenchants. If 160 / Heat Treating of Aluminum and Its Alloys spray quenching is applied on quench-sensitive alloys, a heavy volume spray is needed to almost flood the part. Large flow rates require an abundant water supply. Cost generally dictates the use of a reservoir and recirculating system rather than drawing water from normal supply lines and discharging it after one use. Location of the quench tank or spray chamber with respect to the heat treating furnace is important. To ensure the quench speed generally required, these facilities should be adjacent to the furnace that they service. In elevator-type furnaces, such as the bottom-door or verticaltower, placing the quench tank directly beneath the heating chamber is ideal. Quench tanks should have a ready means of draining and cleaning, for removing contaminants that may discolor the product. Rate of travel of the workpiece through the sprays is an important variable. Local increases in temperature that occur within the first few seconds of quenching, caused by a phenomenon such as plugged spray nozzles, are particularly deleterious. The remaining internal heat may be sufficient to reheat the surface region. When this happens, a large loss in strength occurs at the previously quenched surface. The loss of strength in the affected area of a heavy part is much more severe than that caused by an inadequate quenching rate alone. This is illustrated for 75 mm (3 in.) thick 7075-T62 plate in Fig. 20, which compares, at various depths, the properties of a plate for which quenching was 100 0 0.5 1.0 Depth, in. 1.5 2.0 2.5 3.0 80 70 Longitudinal yield strength, MPa Longitudinal tensile strength, MPa 50 0 Side A 0 600 Control specimen Quenched from side A only Quenched from side B, interrupted after 3 s 10 20 0.5 40 50 30 Depth, mm Depth, in. 1.0 1.5 2.0 60 70 2.5 80 Side B 3.0 80 500 70 60 400 50 300 0 Side A 550 0 10 20 0.5 30 1.0 40 50 Depth, mm Depth, in. 1.5 2.0 60 70 2.5 80 Side B 3.0 70 450 60 350 50 40 250 30 150 0 Side A Fig. 20 10 20 30 40 50 Depth, mm 60 70 Longitudinal tensile strength, ksi 60 Longitudinal yield strength, ksi Hardness, HRB 90 80 Side B Through-thickness property variations due to quench rate and temperature-rise effects in 75 mm (3 in.) thick 7075-T62 plate interrupted on one side after 3 s with those of a plate that was quenched from one side only. Polymer Quenchants Polymer quenchants are widely used in the quenching of aluminum to control distortion and residual stresses. Polymer quenchants retard cooling rates by the formation of films around the part. The effective film coefficient is essentially the heat-transfer coefficient (C), which is related to the Grossmann number (H) as compared with water in Table 2. The application of polymer quenchants is covered in AMS specifications 3025 and 2770, although many aluminum and aerospace companies have developed internal specifications that differ from AMS 2770. Typical parameters for quenching wrought products (other than forgings) in glycol-water solutions are presented in Table 7. For all polymer quenchants, the primary manufacturing variables to achieve a desired quenching rate include: Concentration Agitation Temperature As the concentration of the polymer is increased, the effective quench rate is reduced. As the concentration is increased, a limit will be reached where additions of polymer will not significantly reduce the cooling rate. This concentration is dependant of the molecular weight of the polymer and the type of polymer chosen. Benefits of using aqueous-based polymer quenchant solutions are many fold. Concentrations can be changed quickly and tailored to specific products. There are no fire hazards (in contrast to oil), and an economic recovery system can be put in place to cut the water and chemical costs of operating the systems. Based on experience in the aerospace industry, straightening costs can be reduced by up to 60% compared to using a water quench. Polymer quenchants tend to be more sensitive to agitation. Increasing the agitation Table 7 Limits for quenching in glycolwater solutions Data are for wrought aluminum alloy products other than forgings. Maximum thickness Glycol concentration, vol% Alloys 12–16 17–22 23–28 29–34 35–40 2014, 7075, 2014, 7075, 2014, 7075, 7075, 2014, 2117, 2024, 7075, 2017, 2117, 2024, 7079, 7175, 7178, 2017, 2117, 2024, 7079, 7175, 7178, 2017, 2117, 2024, 7079, 7175, 7178, 7079, 7175, 7178, mm 2219 2.03 7175 25.4 2219 1.80 6061 12.7 2219 1.60 6061 9.53 2219 1.02 6061 6.35 6061 2.03 in. 0.080 1.000 0.071 0.500 0.063 0.375 0.040 0.250 0.080 increases the cooling rate and reduces the polymer film thickness. However, decreasing the agitation can produce nonuniform quenching because of nonuniform film thickness. It also limits the transport of polymer to the part surface. As in every quenching operation, the magnitude and uniformity of agitation is extremely important. Racking of parts is more critical in polymer quenchants because of the strong effects of temperature. Agitation tends to minimize these thermal gradients within the quenchant. The effective quench rate of polymer solutions is affected by temperature. As temperature is increased, the quench rate is reduced. Increasing temperature also increases the oxidation and reduces thermal stability of the polymer, effectively shortening the life of the polymer. The amount of degradation is dependent on the amount of polymer used and the application temperature. Depending on the polymer used, there is also a limit on the bulk quench temperature of the quenchant, because some quenchants will tend to separate or precipitate from solution. This is true of PAG-type polymer solutions. In general, the typical operating temperature range of polymer quenchants is 20 to 40 C (70 to 105 F). Many specifications limit the temperature rise during quenching to 5.5 C (10 F), with a maximum temperature of 43 C (110 F). Polyalkylene Glycol Quenchants Polyalkylene glycol quenchants are the most commonly used polymer quenchants in the heat treating market today (2016). Polyalkylene glycols, or polyalkylene glycol ethers, were first introduced as a family of commercial products in the early 1940s. These materials are formulated by the random polymerization of ethylene and propylene oxides (although higher-alkylene oxides and/or aryl oxides may be used also). Although block polymerizations of these same oxides are possible, these derivatives are less attractive as quenchants. The PAG quenchants are an example of a copolymer. This quenchant is derived from two monomeric units, ethylene oxide and propylene oxide (Fig. 21). By varying the molecular weights and the ratio of oxides, polymers having broad applicability may be produced. Certain of the higher-molecular-weight products were shown to have utility as metal quenchants when used in aqueous solution (U.S. Patent 3,230,893). Proper selection of the polymer composition, and its molecular weight, provides a PAG product that is completely soluble in water at room temperature. However, the selected PAG molecules exhibit the unique behavior of inverse solubility in water, that is, water insolubility at elevated temperatures. This phenomenon provides the unique mechanism for cooling hot metal by surrounding the metal piece with a polymer-rich coating that serves to govern the rate of heat extraction into the surrounding aqueous solution. As the temperature of the metal part approaches the Quenching of Aluminum Alloys / 161 temperature of the quenchant itself (stage C), the PAG polymer coating dissolves to again provide a uniform concentration in the quenchant bath. This is shown in Fig. 22. This mechanism of inverse solubility is limited to two polymer quenchant classes: polyalkylene glycol and polyethyloxazoline. In these systems, as the temperature of the solution is raised, the thermal energy of the system becomes greater than the energy of the hydrogen bond interactions with water. When this occurs, a two-phase system develops, with one layer being water-rich and the other a polymer-rich layer. This is not a clean separation because both phases have some of the other component. The temperature at which this separation occurs is called the cloud point. In PAG quenchants, the ratio of the monomers used to produce PAG quenchants controls the cloud point. In this case, the cloud temperature decreases as the propylene oxide monomer proportion increases. The cloud point also is the basis for the practice of purifying the quenchant bath (see the section “Polyalkylene Glycol Quenchant Reclamation” in this article). In the section describing the cooling characteristics of water, one of the disadvantages cited for plain water is that the vapor blanket stage (stage A) may be prolonged. This prolongation encourages vapor entrapment that may result in uneven hardness and unfavorable distribution of stress, which, in turn, may cause cracking and/or distortion. By using PAG quenchants, uniform wetting of the metal surface results, O thereby avoiding unevenness and the accompanying soft spotting. In fact, selection of the proper PAG quenchant can provide accelerated wetting so that the cooling rates achieved are faster than water and approach those achieved by brines. Thus, brine quenching is possible without the hazards and corrosiveness attendant with the use of salts or caustic solutions. Whereas rusting can be a drawback when quenching with water alone, particularly where recirculation of treated water is not employed, solutions of PAG quenchants may be inhibited to provide corrosion protection of the quench system components. Corrosion inhibition of quenched parts will be of short duration, so that specific protection should be provided following the tempering operation. In AMS 2770, the Aerospace Materials Engineering Committee of SAE has determined the recommended concentrations to be used when quenching aluminum. These concentrations are extremely conservative and are based on a zero-delta strength difference between water and PAG quenchants. They have further categorized the PAG quenchant types into two categories: type I and type II (from SAE International AMS 3025). The physical property differences of these two types of PAG quenchants are shown in Table 8. Allowable concentrations of polymer quenchants for aluminum in accordance with AMS 2770 are shown in Table 9. The influence of polymer concentration on cooling rates is illustrated by the cooling curves O nCH2 – CH2 + mCH2 – CH [(CH2CH2O)n(CH2CHO)m]– CH3 Ethylene oxide Propylene oxide CH3 PAO shown in Fig. 18(b). Cooling curves as a function of PAG concentration are similar to quenching with water at different temperatures (Fig. 18a). The slower rates of cooling achieved at the higher concentrations reflect the thickness of the polymer layer that surrounds the heated part during quenching. The PAG quenchants also are less sensitive to minor changes in polymer concentration, which is a recognized deficiency of polyvinyl alcohol and the other filmforming polymer quenchants. Just as water exhibits a marked decrease in cooling capability as its temperature is elevated (Fig. 15, 18a), this same loss is translated to the aqueous solutions of PAG quenchants. The curves shown in Fig. 23 are illustrative of the general trends that would occur with changes in bath temperature; more detailed data would require specific identification of the particular PAG quenchant employed. In general, low to moderate agitation is essential to ensure that adequate replenishment of polymer occurs at the hot metal surface and to provide uniform heat transfer from the hot part to the surrounding reservoir of cooler quenchant. Figure 24 clearly illustrates that, as agitation is increased, the cooling curves shift to more rapid rates. Polyalkylene glycol quenchants have shown remarkable growth and utility in reducing residual stresses and distortion in aluminum sheet metal, forgings, and castings. The degree of distortion control that can be achieved using PAG quenchants is illustrated in Table 10. Early work (Ref 21) at Boeing using AMS 3025 type I polymer quenchants showed that the use of polymer quenchants substantially reduced the distortion of quenched sheet metal parts. The data (Fig. 25) showed that when quenching 1 mm (0.040 in.) thick aluminum sheet, quench rates exceeding 2700 C/s (4860 F/s) were obtained when quenching in ambient-temperature water. Using a 40% concentration of a type I quenchant, a maximum quenching rate of 1000 C/s (1800 F/s) was observed. This quench rate is Fig. 21 Synthesis of polyalkylene glycol quenchants Fig. 22 Sequence of quenching in a polyalkylene-glycol-type polymer. (a) Moment of immersion; polymer film deposits on component surface. (b) After 15 s, film becomes active. (c) After 25 s, boiling occurs over entire surface. (d) After 35 s, boiling ceases and convection phase begins. (e) After 60 s, polymer starts to redissolve into solution. (f) After 75 s, film has completely redissolved and heat removal is entirely by convection. Courtesy of Houghton International 162 / Heat Treating of Aluminum and Its Alloys approximately 10 times the rate necessary to achieve full properties in aluminum sheet. Further work (Fig. 26) showed significant reductions in distortion as the concentration of type I quenchant was increased. Additional work at Northrup (Ref 22) showed that sheet metal products could be quenched in polymer concentrations up to 40%. When quenching similar sheet metal parts in ambient water, quenching rates through the critical range of 400 to 300 C (750 to 570 F) exceeded 2200 C/s (3960 F/s), significantly higher than the 100 C/s (180 F) necessary to achieve full quenching. The results of this work are shown in Fig. 27. A dramatic visual demonstrating the benefits of PAG quenchants in controlling distortion is shown in Fig. 28. Often, large castings and forgings are quenched in hot water to achieve residual-stress control. Often, these quench rates are slow and properties are diminished; however, the residualstress and distortion concerns outweigh property considerations. It is apparent that the cooling rates of PAG quenchants are similar at high concentrations to that of elevated-temperature water (Fig. 18). Concentrations of up to 60% type I quenchant achieved similar quench rates as a 93 C (200 F) water quench (Ref 23, 24). These data (Fig. 29) provide for proper selection of PAG concentrations for large forgings and castings, provided that reference data on alloy and properties can be determined for elevated-temperature water quenching. Cooling curve analysis of polymer quenchants is more dependent on agitation than are oil quenchants. To overcome this, two standards have been developed to accurately measure the cooling curves of polymer quenchants: ASTM D6482 (Tensi method) and ASTM D6549 (Drayton method). Each method provides for a different method of agitation of the quenchant. The results of the tests cannot be compared to each other. Many auditing agencies (National Aerospace and Defense Contractors Accreditation Program, CQI-9 Heat Treatment System Assessment, etc.) are requiring cooling curve analysis of polymer quenchants on a monthly or quarterly basis. Table 8 Physical properties of polyalkylene glycol quenchants in accordance with AMS 3025 Neat Diluted to 20% Property Type I Type II Water content, neat % Specific gravity Refractive index Viscosity at 38 C (100 F), cSt Viscosity at 38 C (100 F), cSt Cloud point, C ( F) 45–48 1.094 ± 0.005 1.4140 ± 0.005 535 ± 70 5.5 ± 0.5 165 ± 5 (330 ± 9) 57–63 1.080 ± 0.025 1.3910 ± 0.005 300 ± 20 4.4 ± 0.5 165 ± 5 (330 ± 9) Table 9 Limits for quenching in polymer solutions in accordance with AMS 2770H Maximum thickness(b) Polymer type(a) Alloy I 2024 Sheet, extrusions 2219 6061 Sheet, extrusions Sheet, plate, bar, extrusions 7049 7050 7075 Sheet, plate, bar 6061 7075 Forgings 7049 7149 Forgings 7050 Forgings 7049 7050 7075 2024 Extrusions II mm in. Polymer concentration (a)(c), % 1.02 1.60 1.80 2.03 1.85 6.35 9.52 25.4 2.03 6.35 9.52 12.70 25.4 25.4 50.8 63.5 25.4 50.8 76.2 25.4 50.8 76.2 101.5 6.35 9.52 0.040 0.063 0.071 0.080 0.073 0.250 0.375 1.00 0.080 0.250 0.375 0.500 1.00 1.00 2.00 2.50 1.00 2.00 3.00 1.00 2.00 3.00 4.00 0.250 0.375 34 max 28 max 22 max 16 max 22 max 40 max 32 max 22 max 40 max 34 max 28 max 22 max 16 max 18–22 11–15 8–12 18–22 11–15 8–12 28–32 24–28 18–22 13–17 28 max 22 max (d) (d) (d) (d) (d) ... 1.02 1.60 2.03 1.02 4.83 6.35 0.040 0.063 0.080 0.040 0.190 0.250 34 22 16 34 20 18 (d) (d) (d) ... 1.0 2.0 11– 15 8–12 Form Sheet, extrusions Sheet, plate, bar 6061 7049 7050 7075 6061 7075 Forgings 25.4 50.8 Notes ... ... (e) (e) ... ... ... max max max max max max ... (e) (a) Types I and II polymer solutions and concentrations shall conform to AMS 3025. Concentrations are percentages by volume of the undiluted polymer as furnished by the producer. (b) Thickness is the minimum dimension of the largest section at the time of heat treatment. (c) Where only maximum concentration is shown, any 4% range may be used, except the maximum shown shall not be exceeded. When concentration is specified on a drawing or purchase order without tolerance or range, the tolerance shall be ±2%. (d) Applicable when final temper is T4 or T42. When final temper is T6 or T62, sheet and plate up to 6.35 mm (0.250 in.), inclusive, may be quenched in types I or II polymer solution at 22% max. (e) Prohibited for 7075 alloy over 25 mm (1 in.) when final temper is T6 35 70 110 Cooling rate, °F/s 145 180 215 250 325 360 35 900 1470 800 1290 700 3 2 1 600 1110 500 930 400 750 Curve 1: 20 °C Curve 2: 40 °C Curve 3: 60 °C 300 20 Fig. 23 40 60 80 100 120 140 Cooling rate, °C/s 290 325 360 1650 1470 2 1 1290 3 4 1110 930 Flow rate m/s Curve 1: Nil Curve 2: 0.8 Curve 3: 1.6 Curve 4: 2.4 300 100 200 Influence of temperature on cooling curves of a polyalkylene glycol quenchant. Courtesy of Houghton International Cooling rate, °F/s 145 180 215 250 400 Concentration: 25% 212 Agitation: Vigorous 180 110 500 200 160 70 600 390 200 100 570 Temperature, °C 1650 800 Temperature, °F 900 700 Temperature, °C 290 570 390 Concentration: 25% Temperature: 40 °C 20 Fig. 24 750 40 60 80 100 120 140 Cooling rate, °C/s 160 180 Temperature, °F Condition 212 200 Influence of agitation on the cooling curves of a polyalkylene glycol quenchant. Courtesy of Houghton International Quenching of Aluminum Alloys / 163 Polyalkylene Glycol Bath Maintenance industrial environment will eventually be contaminated with dirt and debris from the parts that are being heat treated. This, of course, includes scale. Quench performance over time will be affected by alterations of the total chemistry of the bath by these impurities. Filtering the solutions with cartridge or bag filters using 5 to 10 mm filter media has proven sufficient to keep the bath in a condition where concentration measurements are accurate. Bath conditioning also consists of controlling the bioburden in the bath. Concentration control is accomplished using densitometers, refractometers, and viscosity meters. Several of the instruments require frequent calibration, which adds to the maintenance burden in the factory. The refractive index monitor with remote sensing and optional connection to a PLC has proven to be very stable if the solution is conditioned and filtered. Obtainable accuracy levels over time are within ±0.5%. The refractive index of PAG polymer solutions (in the range employed for quenching) is essentially linear with concentration (Fig. 30). Thus, the refractive index of a PAG quenchant solution serves as a measure of product concentration. Industrial model optical refractometers that employ an arbitrary scale may be cali- To be effective and economical, aqueous polymer solutions require proper maintenance. The ability to obtain better and more controlled quench rates is a major factor in deciding to implement a PAG quench into a production process. Proper control and performance of these solutions involves concentration control and bath conditioning. Reclamation also is an important economic consideration. The concentration of the polymer in the quench bath has one of the most significant influences on the finished product. The cleanliness of the bath directly influences the accuracy of the measurements. With the use of a programmable logic controller (PLC) and operator interfaces, concentration changes tailored to the product can be carried out accurately and quickly. The use of fully automatic systems has proven somewhat impractical because troubleshooting becomes difficult. For example, the status of the filling and draining operations is hard to monitor. Semiautomatic systems, where each phase is initiated by an operator, have proven more robust and less troublesome. Bath conditioning consists of filtering and tending to the control of any biological impurities in the solutions. Any bath that is used in an Table 10 Degree of distortion control achieved using polyalkylene glycol (PAG) quenchants in aluminum Distortion in PAG quenchants Distortion in water Part type Die forging Die forging Machined bar Dip-brazed chassis Alloy mm 7075-T6 2014-T6 7075-T6 6061-T6 2.5–3.8 in. mm 0.10–0.15 0.05 in. 0.002 Unknown 10 None 0.40 Unknown 0.075 0.08 0.003 0.003 Source: Ref 20 1800 3600 Cooling rate, °F/s 5400 7200 9000 10,800 12,600 Air LN2 Other Other 0 40 20 LN2 40 PAG in water at 26 °C, % 14 11 8 6 4 Water temperature, °C Air 17 2 56 30 20 11 6 2 25 10 0 1000 2000 3000 4000 Cooling rate, °C/s 5000 6000 7000 Effect of type I polyalkylene glycol (PAG) concentration on the maximum cooling rate when quenching 1 mm (0.04 in.) thick 2024 aluminum sheet. Source: Ref 21 Water Quenchant Type I PAG 26 °C, % 22 Fig. 25 brated. Whereas such instruments prove invaluable for day-to-day monitoring of the quenchant concentration, the refractometer also will register other water-soluble components that are introduced to the used quenchant. When the indicated refractometer reading begins to provide erroneous numbers, some other analytical test is required to define the effective quenchant concentration. With PAG quenchants, kinematic viscosity measurements (which are correlated with concentration) have proven to be most useful. These plots also are available from quenchant manufacturers and are similar to those for the refractive index, with one exception: The viscosity usually has a nonlinear relationship to the polymer concentration. Therefore, firstorder linear regression analysis should not be used as a line-fitting procedure. Examples of kinematic viscosity versus concentration are shown in Fig. 31 and 32 for types I and II PAG quenchants. Biological Contamination and Control. Because PAG quenchants are, for the most part, resistant to bacteria and fungi, the addition of a bactericide to the as-supplied quenchant is not required. However, growth of microorganisms can occur in use when contaminants (such as oils) act as a nutrient. Cleanliness and oxygen content are the keys. Solutions should remain agitated to minimize available sources of nutrients; rust and other solids are typical food sources. Anaerobic bacteria and fungi thrive in oxygendepleted environments. Stagnant solutions contribute to localized oxygen depletion; thus, quenchants should be kept moving to prevent oxygen depletion. Additional bubbling of air can be used, but it is generally most effective to continue agitating the quenchants during shutdowns. Anaerobic bacteria and fungi can breed in typical polymer quench tanks. These bacteria and fungi are not usually a health hazard but an 26 °C 0 Fig. 26 2 4 6 8 Distortion, % 10 12 14 Effect of type I polyalkylene glycol (PAG) concentration on the distortion of 1 mm (0.04 in.) thick 2024 aluminum sheet. Source: Ref 21 164 / Heat Treating of Aluminum and Its Alloys odor issue. These bacteria contribute to “Monday morning smells” when equipment is first turned on after a weekend of sitting stagnant. Aqueous solutions will experience bacteria and algae growth if there are no biocides present. Bacteria growth can cause corrosion of parts (microbiologically induced corrosion) and can detrimentally affect membranes used for separation in reclamation systems. The bacteria can also reduce the sodium nitrate in the Cooling rate (400–300 °C), °C/s 0.51 10 0.81 3 1800 1.02 1.80 3.18 Cooling rate (750–570 °F), °F/s 18,000 104 180 102 0 5 10 15 20 25 30 35 40 Type I quenchant, % Fig. 27 Effect of type I polyalkylene glycol concentration on quenching rates through the critical range of 400 to 300 C (750 to 570 F) on different thicknesses of sheet metal (0.50 to 3.2 mm, or 0.02 to 0.13 in.) Fig. 28 Sheet metal quenched in ambient water and 20% type I polyalkylene glycol (PAG). (a) Water-quenched sheet metal. (b) Identical sheet metal panels quenched in 20% type I PAG. Courtesy of Houghton International 103 bath if they are anhydrous bacteria. Algae will coat the insides of the tanks and piping and result in incorrect concentration data. Biocides are used with various successes. A properly designed filtration system, with adequate turnover, can help minimize bacteria and fungi problems. Sand (swimming pool) filters are very effective and are capable of filtering to 6 to 8 mm. Bag and cartridge filters can be a breeding ground for fungi and bacteria because of low fluid flows and a high concentration of food sources. Should fungi and bacteria become a problem, then effective treatment is necessary by the application of the appropriate biocide. Microbiological treatment, such as is employed with other aqueous metal working fluids, generally can keep biological activity under control. The polymer quenchant supplier can offer assistance in the selection of the proper biocide and the amount to be added. Usually, a large kill dose is recommended. Constant application of biocides can contribute to resistant bacteria. In the event that this occurs, large kill doses and switching biocides on a routine basis are recommended. Because biocides are designed to kill living organisms, it is absolutely critical that proper personal protective gear be used. Refer to the biocide suppler for proper recommendation regarding dosage and protective equipment. Biocides with glutealdehyde are the most commonly used. They last from 10 to 21 days in the bath and must be replenished periodically to remain effective. Shop test procedures that check for bacteria and fungi will tell the operators of the need to treat the bath. Small paddle sticks are used for this testing, with satisfactory results. An occasional change of biocide will keep the bacteria from becoming resistant to the product. Contamination is a common occurrence in the heat treating shop. Hydraulic fluids and solids such as soot and rust are common. Sediment such as scale and soot can hinder concentration control by making the refractive index hard to measure. These contaminants change 45 1800 180 25.4 50.8 10 18 76.2 1.8 35 30 Concentration, % 10 12.7 2 Cooling rate (750–570 °F), °F/s Cooling rate (400–300 °C), °C/s 40 25 20 15 10 5 1 0 5 10 15 20 25 Type 1 PAG concentration, % 30 35 40 0 0 5 10 15 Fig. 29 Interrelationship of part thickness and type I polyalkylene glycol (PAG) concentration for forgings and castings from 12.5 to 75 mm (0.5 to 3.0 in.) thickness. Source: Ref 23 20 25 Brix, degrees Type I Fig. 30 Type II Relationship of refractive index, measured by Brix refractometers, on the concentration of types I and II polyalkylene glycol quenchants 45 45 40 40 35 35 30 30 Concentration, % Concentration, % Quenching of Aluminum Alloys / 165 25 20 15 25 20 15 10 10 5 5 0 0 Fig. 31 5 10 15 Kinematic viscosity, cSt 20 25 Kinematic viscosity versus concentration for type I polyalkylene glycol quenchants the cooling curve behavior. The effect of each of these contaminants is site and process specific. In general, excessive contamination can be eliminated through proper filtration and skimming of oils and other contaminants. In the case of PAG quenchants, thermal separation and the readdition of appropriate corrosion inhibitors can clean the polymer solution. Chemical Control. The basic chemistry of new PAG changes very little over time when used in the quench bath. The pH level can change and must be maintained by adding buffers in accordance with the manufacturers’ recommendations. The use of reverse osmosis (RO) membranes requires special modifications to the PAG. Mainly, the pH value is lowered slightly to increase membrane life. However, the pH must not be lowered too much, because the PAG becomes unstable at pH values less than 6 to 6.5. A low pH value can cause corrosion of aluminum parts during quenching. The corrosion inhibitor used in PAG is commonly sodium nitrate. This salt will be depleted over time and must be replenished to protect piping, pumps, and other equipment. Sodium nitrate is also one of the first products that migrates through a worn RO membrane and creates high electrical conductivity in the permeate water. This can be used as an indicator for tracking the condition of the membranes over time. Polyalkylene Glycol Quenchant Reclamation With the development of fully closed loop systems with variable concentration control and conditioning, the costs of PAG replacement have drastically decreased in comparison to previous practice when rinse water was flushed to a drain, causing dragout from the quench tank to be lost. The capital cost of installing these systems must be compared to the savings in PAG replacement cost. Wastewater reduction also can be a major factor in some regions. 0 0 Fig. 32 2 4 6 8 10 12 Kinematic viscosity, cSt 14 16 18 Kinematic viscosity versus concentration for type II polyalkylene glycol quenchants There are basically three ways of separating PAG from the water after it has been diluted to the concentration needed by the user: Heat separation Membrane separation using micro- or nanofiltration Membrane separation using RO technology The cost of maintaining the systems has decreased. This is particularly true with RO systems, because third-generation control and hardware are now in place. These have proven to be reliable over time. Heat Separation. As noted, the cloud point phenomenon can be used as a way to purify the quenchant bath. The temperature of the bath is increased until separation occurs. The tank is heated to approximately 75 to 85 C (165 to 185 F), and the PAG will settle out to the bottom, unless there are considerable amounts of salt present, in which case it settles to the top. After separation and the polymer settling on the bottom of the tank, water is pumped or siphoned from the tank. Fresh water is then added to the desired concentration. Inorganic rust inhibitors are usually present as part of the water phase and also must be added to the system to retain the proper corrosion inhibition. One additional consideration is that the increased temperature will tend to oxidize the polymer, resulting in shorter life. Membrane Separation Using Micro- or Nanofiltration. This method uses membranes that allow the water to pass but not the PAG and salts, which stay on the process side of the membranes. However, small amounts of PAG will pass through the membranes with the water, resulting in some waste of PAG. The PAG also will break down over time to smaller molecules as a result of mechanical and thermal action on the polymer. Membrane Separation Using RO Technology. Figure 33(a) illustrates a typical closedloop RO system. With this method, PAG is separated from the water using membranes that allow the water to pass but reject the PAG and salt, which stay on the process side of the membranes. The water (permeate) is stored in a water tank for later use or sent to drain. This technology does not work well in conjunction with salt baths or steel heat treating because the salt concentration in the PAG will increase during the concentration cycle. Salt is not desired in the quench bath because it can cause corrosion on the parts. Steel scale and free iron will damage the membranes and must be removed from the solution before it reaches the RO machine. A new separation method has been developed that uses the heat separation concept, and it requires only a single pass-through. Figure 33(b) shows the schematic for this system. Note that the process tank is optional compared to the RO system shown in Fig. 33(a). The heat separation method does not use membranes and is not sensitive to salt or iron in the bath. Production testing concentrated a 1% PAG solution into a 60% solution and clean water in one pass at a rate of 3.8 L/min (1 gal/min). Other concentrations included 22% PAG, where the recovery rate also proved to be up to 60% PAG on the product side of the stream and clean water on the other side. The system is very compact, robust, and less costly than RO separation. At this time (2016), the method is implemented in several locations. Other Quenching Media Air Quenching. For maximum dimensional stability, some forgings and castings are fan cooled or still-air cooled. In such instances, precipitation-hardening response is limited, but satisfactory values of strength and hardness are obtained. Alloys that are relatively dilute, such as 6063 and 7005, are particularly well suited to air quenching, and their mechanical properties are not greatly affected by its low cooling rate. Lower quenching rates are also employed for forgings, castings, and complex 166 / Heat Treating of Aluminum and Its Alloys Fig. 33 Membrane separation of polyalkylene glycol (PAG) and water with (a) closed-loop reverse osmosis system and (b) one-pass heat separation. Courtesy of Bogh Industries shapes to minimize warpage or other distortion and the magnitude of residual stresses developed as a consequence of temperature nonuniformity from surface to interior. Certain alloys that are relatively insensitive to cooling rate during quenching can be either air cooled or water quenched directly from a final hot-working operation. In either condition, these alloys respond strongly to precipitation heat treatment. This practice is widely used in producing thin extruded shapes of alloys 6061, 6063, 6463, and 7005. Upon precipitation heat treating after quenching at the extrusion press, these alloys develop strengths nearly equal to those obtained by adding a separate solution heat treating operation. Quenching in Nitrogen. There has been limited interest in quenching with nitrogen. The motivation was that the extremely cold temperatures of liquid nitrogen could provide a rapid quench rate. However, with vaporization of liquid nitrogen in contact with the hot part, the quenchant medium is actually nitrogen vapor around the part, and the vapor pocket does not collapse even with reasonable agitation (Ref 1). Thus, nitrogen is very limited to quench-insensitive alloys, such as thin gages of 6061 sheet or very thin (<0.75 mm, or 0.030 in.) gages of 2024. Fast Quenching Oils. With the advent of polymer quenchants, quenching oils are rarely used. Prior to polymer quenchants, oil quenchants were only used in rare instances to control distortion for castings of certain thicknesses. Fast quenching oils were required for aluminum alloy quenching. Fluidized-Bed Quenching. Recently, there has been interest in using fluidized-bed technology for the solution heat treatment of aluminum alloys (Ref 25, 26). A fluidized bed consists of a medium of fine, hard particles (i.e., sand) that is partially suspended by a fluidizing gas. The partial suspension of the medium allows the particles to easily slide past each other, resulting in the fluidizing bed acting very similar to a fluid. The fluidlike nature of the fluidized bed allows for easy insertion, conveyance, and extraction of parts for heat treating. Fluidized-bed technology has found wide use in the heat treatment of steels, but its use for heat treating other metals has been limited. In the heat treatment of aluminum alloys, fluidized-bed quenching is an attractive alternative to liquid quenching processes because the part does not develop a vapor barrier during quenching. This lack of a vapor barrier can significantly reduce residual stresses and part distortion. Compared to quenching in water, it was shown that quenching in the fluidized bed reduced residual stresses by nearly 70% in an A356.2 casting (Ref 27). The heat-transfer rate of fluidized-bed quenching is intermediate between that of water and air quenching (Fig. 34). The lower heat-transfer rate limits fluidized-bed quenching to thinner sections or alloys with a low quench sensitivity, such as 319 casting alloy. Fig. 34 Comparison of heat-transfer coefficient for water, forced-air, and fluidized-bed quenching precipitation that occurs during cooling, and the method of quench-factor analysis also provides a method of correlating cooling curves with quantitative prediction of properties. Quench-factor analysis is based on the principle of additivity when a precipitation process is isokinetic, where transformation rates only differ by a rate constant according to the Avrami equation: hti d ¼ 1 exp k Quench-Factor Analysis Although average quench rates through a critical temperature range can provide reasonable property predictions if cooling rates are fairly uniform, cooling rates typically vary considerably during the quench. Isothermal timetemperature-transformation (TTT) diagrams, although useful to define critical times for a specified amount of precipitation at a given temperature, do not quantify the overall extent of precipitation during continuous cooling. Quench-factor analysis is a method of quantifying the extent of precipitation after continuous cooling from the isothermal C-curve and either an experimental or analytical cooling curve. The properties of aluminum alloys also are dependent on the amount of alloy where d is the fraction of precipitation that occurs in time t during the quench, and k is a temperature-independent constant. The value of k depends on the degree of supersaturation and the rate of diffusion from the study of transformation during continuous cooling processes using isothermal kinetics by Avrami (Ref 28, 29). Subsequently, Cahn demonstrated that the precipitation process during continuous (nonisothermal) cooling is additive for isokinetic transformation, such that (Ref 30): Z tf t¼ ti dt CT (Eq 12) where t is the measured amount of transformed product (also designated as Q in quench-factor Quenching of Aluminum Alloys / 167 analysis), t is the time from the cooling curve, ti is the time at the start of the quench, tf is the time at the end of the quench, and CT is the critical time from the TTT C-curve. When t (or Q) = 1, the fraction transformed equals the fraction represented by the isothermal C-curve. Therefore, the continually cooled material will exhibit the same behavior as one quenched instantaneously to an intermediate temperature, held for the critical time at that temperature, and quenched instantaneously to room temperature. From these basic relationships, Evancho and Staley (Ref 3) developed a method of quenchfactor analysis that quantitatively calculated the extent of precipitation and the properties from a cooling curve and the critical times (CT) along the isothermal C-curve. They defined the constant, k, in the Avrami equation relative to the critical time, such that: a nucleus, k4 is a constant related to the solvus temperature, k5 is a constant related to the activation energy for diffusion, R is the gas constant equal to 8.3143 J K1 mol1, and T is the temperature in Kelvin. Numerical values for each of these constants define the C-curve for the particular alloy composition and temper condition, and published coefficients for the CT values of various alloys are listed in Table 11. With a given cooling curve and the isothermal C-curve of a given alloy, the quench-factor calculation basically involves the summation of incremental quench factors over a sequence of time intervals, as illustrated in Fig. 35. The incremental quench factor (qi) over a small time interval (△ti) is: k ¼ CT =k1 where CTi is the critical time defined as the time difference between the cooling curve where k1 is a constant that equals the natural logarithm of the fraction untransformed (i.e., 1 minus the fraction defined by the C-curve); if 0.5% is untransformed, then k1 = ln[0.995] = 0.0050. Moreover, because the properties of aluminum alloys also are dependent on the amount of alloy precipitation that occurs during cooling, the k1 constant based on the specified fraction of untransformed product also provides a specified fraction of a property (such as 99.5% of attainable yield strength after aging with k1 = 0.0050). In their 1974 paper, Evancho and Staley showed that from the Avrami equation, the attainable strength is a function of the amount of solute remaining in solution after the quench and is related to k1: k1 ¼ ln sx smin smax smin qi ¼ Dti CTi of the quench process and the isothermal Ccurve (Fig. 36). Over the entire cooling curve, the total quench factor (Q) is thus the summation of incremental quench factor as follows: Q¼ Dt final X The cumulative quench factor (Q, or t in Eq 12) reflects not only the precipitation kinetics of the alloy but also the cooling rates (which are a function of quenchant fluid, quenchant velocity, temperature, part thickness, and other variables). An alloy with a low rate of precipitation will produce a lower Q value at a given cooling rate compared to an alloy with a high precipitation rate. The overall extent of precipitation (and thus property development) also can be defined by quench factors for a given alloy and temper. For example, the quench factors for alloy 7075-T63 in Table 12 list the levels Table 11 Coefficients for calculating quench factors at 99.5% of attainable yield strength Calculated range k1(a) Alloy 7010-T76 7050-T76 7075-T6 7075-T73 7175-T73 2017-T4 2024-T6 2024-T851 2219-T87 6061-T6 356-T6 357-T6 Al-2.7Cu-1.6Li-T8 0.00501 0.00501 0.00501 0.00501 0.00501 0.00501 0.00501 0.00501 0.00501 0.00501 0.0066 0.0062 0.0050 k2 , s 5.6 2.2 4.1 1.37 1.8 6.8 2.38 1.72 0.28 5.1 3.0 1.1 1.8 1020 1019 1013 1013 109 1021 1012 1011 107 108 104 1010 108 k3, J/mol k4, K 5780 5190 1050 1069 526 978 1310 45 200 412 61 154 1520 897 850 780 737 750 822 840 750 900 750 764 750 870 k5, J/mol 1.90 1.8 1.4 1.37 1.017 2.068 1.47 3.2 2.5 9.418 1.3 1.31 1.02 105 105 105 105 105 105 105 104 104 104 105 105 105 CT ¼ k1 k2 exp R T ðk4 T Þ2 ! C 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 425–150 F Ref 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 800–300 31 3 3 32 33 32 34 35 36 32 37 37 37 (a) k1 is a unitless value that corresponds to the unprecipitated fraction. For this analysis, it is usually 0.995 and ln[0.995] = 0.00501. Source: Ref 38 with k1 = 0.0050, then sx = 0.995 smax. With these physical principles relating the extent of precipitation (and also correlation with properties associated with the extent of precipitation during continuous cooling), Evancho and Staley determined the best fit equation for critical time (CT) values from multiple linear regression analysis of empirical C-curves for 7050 and 7075 aluminum alloys. The resulting equation for critical time (CT) along an isothermal C-curve developed by Evancho and Staley is: k3 k42 qi Dt1 k5 exp RT where CT is the critical time required to precipitate a constant amount of solute (the locus of the critical times is the C-curve), k1 is a constant that equals the natural logarithm of the fraction untransformed during quenching (i.e., the fraction defined by the C-curve, typically ln[0.99] or ln[0.995]), k2 is a constant related to the reciprocal of the number of nucleation sites, k3 is a constant related to the energy required to form Fig. 35 Method of determining quench factor Q using a cooling curve and a C-curve 168 / Heat Treating of Aluminum and Its Alloys Table 12 Relationship between quench factor and yield strength in 7075-T73 Quench factor (Q) Fig. 36 Illustration of incremental quench-factor coefficients with cooling curve superimposed on the C-curve of an isothermal transformation diagram. Source: Ref 15 of obtainable yield strength after aging. These quench factors provide a quantitative way of evaluating the necessary cooling curve to obtain the required yield strength, or the quench factors could be used to consider suitable properties with lower quench rates. Yield strength prediction from quench-factor analysis is much more accurate than predictions from average cooling rates (Table 13). Yield strengths predicted from quench factor agree very well with measured yield strengths for all specimens, the maximum error being 19.3 MPa (2.8 ksi). Yield strengths predicted from average quenching rates, however, differ from measured values by as much as 226 MPa (32.8 ksi). The advantage of using the quench factor for predicting yield strength from cooling curves is apparent. In addition, quenchfactor analysis is particularly advantageous in evaluating the property effects of cooling curves that have holding times either above or below the critical temperature range. With an upper limit quench factor defined, the appropriate quenching process and cooling rate in the section of interest can be determined. This method of describing quench severity is different from that used with Grossman numbers (H), which are related solely to the ability of a quenchant to extract heat and not to the transformation kinetics of the alloy being heat treated. However, an underlying assumption of both quench-factor analysis and averagecooling-rate estimation is that the only effect of temperature is on the kinetics of precipitation. This assumption is not valid, however, when portions of the metal are quenched locally but reheated significantly before quenching is complete. More information on the application of quench-factor analysis is in the article “Quench Sensitivity of Aluminum Alloys” in this Volume. Residual Stress and Distortion. Aluminum is extremely prone to distortion during heat treatment and quenching. Residual stresses and distortion originate from temperature gradients, and large thermal strains are developed within and across the surface of parts during quenching or solution heat treatment of aluminum alloys. This can contribute to large-part distortion and residual stresses (Fig. 37). Of the three basic steps involved in the heat treating of aluminum alloys (solution heat treating, quenching, and aging), the most severe distortion is usually observed after quenching from the solution heat treating. In most cases, distortion occurs because the quench is too severe. However, another important factor is racking practice. Proper racking of parts, as described in the next section of this article, is a very important factor for controlling distortion, both in the furnace and during the quench. During the quench, residual stresses and distortion originate from temperature gradients. The gradient induces plastic deformation from contraction or expansion in the part. Because the surface of the part cools first, it tends to contract, thereby imposing a state of compressive stress on the interior. The reaction places the surface in tension. The surface layer deforms plastically when the tensile stress exceeds the flow stress of the material. Then, as the interior of the part cools, it is restrained from contracting by the cold surface material. The resulting reaction places the surface in a state of compressive stress and the center in a state of tensile stress. When the part is completely cooled, it remains in a state of equilibrium, with the surface under high compression stresses balanced by tensile stresses in the interior. Generally, the compressive stresses in the surface layers of a solid cylinder are two-dimensional (longitudinal and tangential), and the tensile stresses in the core 0.0 2.0 4.0 6.0 8.0 10.0 12.0 14.0 16.0 18.0 20.0 22.0 24.0 26.0 28.0 30.0 32.0 34.0 36.0 38.0 40.0 42.0 44.0 46.0 48.0 50.0 Predicted yield strength Attainable yield strength, % MPa ksi 100.0 99.0 98.0 97.0 96.1 95.1 94.2 93.2 92.3 91.4 90.5 89.6 88.7 87.8 86.9 86.0 85.2 84.3 83.5 82.7 81.8 81.0 80.2 79.4 78.6 77.8 475.1 470.2 465.4 461.3 456.5 451.6 447.5 442.7 438.5 434.4 429.6 425.4 421.3 417.2 413.0 408.9 404.7 400.8 396.5 393.0 388.9 384.7 381.3 377.2 373.7 369.6 68.9 68.2 67.5 66.9 66.2 65.5 64.9 64.2 63.6 63.0 62.3 61.7 61.1 60.5 60.0 59.3 58.7 58.1 57.5 57.0 56.4 55.8 55.3 54.7 54.2 53.6 are triaxial (longitudinal, tangential, and radial), as illustrated in Fig. 38. The magnitude of the residual stresses is directly related to the temperature gradients generated during quenching. Conditions that decrease the temperature gradient reduce the residual-stress ranges (Ref 39). Quenching variables that affect the temperature gradient include the temperature at which quenching begins, cooling rate, section size, and variation in section size for nonflat products. For a part of a specific shape or thickness, lowering the temperature from which the part is quenched or decreasing the cooling rate reduces the magnitude of residual stress by reducing the temperature gradient. Figures 39 and 40 illustrate the effect of quenching temperature and cooling rate, respectively. With a specific cooling rate, the temperature gradient is greater in a section of large diameter or thickness than it is in a smaller section. Therefore, the residual stresses in the larger section are higher (Fig. 41). In products having differences in cross section, large temperature gradients can be minimized by covering or coating the thinner sections with a material that decreases the quench rate, so that it more closely matches that of the thicker sections. The range of residual stresses generated during quenching varies considerably for different alloys. Those properties related to alloy composition that specifically affect the thermal gradient and the degree of plastic deformation that occur during quenching are involved. High residual stresses are promoted by high values of properties such as Young’s modulus of elasticity, proportional limit at room and elevated Quenching of Aluminum Alloys / 169 Table 13 Yield strength values for 7075-T6 sheet predicted from cooling curves using average quench rate and quench factor Average quench rate from 400 to 290 C (750 to 550 F) Measured yield strength Yield strength predicted from average quench rate Yield strength predicted from quench factor Quench F/s Quench factor, t (or Q) MPa ksi MPa ksi MPa ksi Cold water Denatured alcohol to 290 C (550 F), then cold water Boiling water to 315 C (600 F), then cold water Still air to 370 C (700 F), then cold water 935 50 1680 90 0.464 8.539 506 476 73.4 69.1 499 463 72.4 67.2 498 478 72.3 69.4 30 55 15.327 458 66.4 443 64.2 463 67.1 5 9 21.334 468 67.9 242 35.1 449 65.1 C/s Fig. 39 Fig. 38 Residual-stress diagram for 2014 alloy quenched in cold water from 500 C (935 F) temperature, the coefficient of thermal expansion, and by a low value of thermal diffusivity. These property factors affect the magnitude of residual stresses to different degrees. The Fig. 37 Distorted 7050 wing spar improperly racked, resulting in excessive distortion and scrap Effect of quenching temperature on residual stress in 5056 alloy cylinders 76 by 229 mm (3 by 9 in.) quenched in water at 24 C (75 F) influences of the coefficient of thermal expansion and elevated-temperature yield strength are especially significant. For example, a low coefficient of thermal expansion can counteract a high proportional limit. The net effect is a low residualstress level, or an alloy such as 2014 can develop a high residual-stress range because of its very high elevated-temperature strength, despite average values for coefficient of thermal expansion, modulus of elasticity, and thermal diffusivity. The effects of residual stresses from quenching require consideration in the application of heat treated parts. Where the parts are not machined, the residual compressive stresses at the surface may be favorable by lessening the possibility of stress corrosion or initiation of fatigue. However, heat treated parts are most often machined. Where the quenching stresses are unrelieved, they can result in undesirable distortion or dimensional change during machining. Metal removal upsets the balance of the residual stresses, and the new system of stresses that restores balance generally results in warpage of the part. Further, in the final balanced stress system, the machined surfaces of the finished part can be under tensile stress with attendant higher risk of stress corrosion or fatigue. Because of the practical significance of residual stress in the application of heat treated parts, various methods have been developed either to minimize the residual stresses generated during quenching or to relieve them after quenching. The methods commonly used for stress relieving heat treated parts include mechanical and thermal. The methods used to avert the development of high residual stresses during quenching rely on a reduced cooling rate to minimize the internal or external temperature gradients. Using quenching media that provide less rapid cooling during quenching of irregular-shaped parts is common practice. For this reason, the quenching of large die forgings and castings in hot (60 to 80 C, or 140 to 175 F) or boiling water is a common practice. However, quench-rate-sensitive alloys may 170 / Heat Treating of Aluminum and Its Alloys Fig. 40 Effect of quenching rate on residual stresses in 2014 and 355 alloy cylinders 75 mm by 230 mm (3 by 9 in.) quenched from 500 and 525 C (930 and 975 F), respectively Fig. 41 Effect of section size on residual stress in 2014 cylinders quenched from 505 C (940 F) in water at 20 C (70 F) suffer a loss in mechanical and corrosion properties due to solute loss. Intergranular corrosion resistance may also be impaired by these reduced cooling rates. Polyalkylene glycol (10 to 40% in water) is effective as a quenchant in minimizing residual stresses and distortion with little loss in properties. Due to inverse solubility, a film of the liquid organic polymer is immediately deposited on the surface of the hot part when the part is immersed in the quenchant. By reducing the rate of heat transfer, the deposited film reduces thermal gradients. One approach to reducing the cooling rate differential between surface and center is the use of a milder quenching medium, such as water that is hotter than that normally used or a water-PAG polymer solution. Boiling water, which is the slowest quenching medium used for thick sections, is sometimes employed for quenching wrought products, even though it lowers mechanical properties and corrosion resistance. Quenching of castings in boiling water, however, is standard practice and is reflected in design-allowable properties. Another approach to minimizing residual stresses that is generally successful consists of rough machining to within 3.2 mm (0.125 in.) or less of finish dimensions, heat treating, and then finish machining. This procedure is intended to reduce the cooling-rate differential between surface and center by reducing thickness; other benefits that accrue if this technique is used to reduce or reverse surface tension stresses in finished parts are improvements in strength, fatigue life, corrosion resistance, and reduced probability of stress-corrosion cracking. Several factors (especially quenching warpage) sometimes preclude general use of this procedure. The thinner and less symmetrical a section, the more it will warp during quenching, and the residual stresses resulting from straightening of warped parts (plus straightening costs) often are less desirable than the quenching stresses. Holding fixtures and die quenching may be helpful, but precautions must be taken to ensure that they do not excessively retard quenching rates. Other factors that must be considered are the availability of heat treating facilities and whether or not the advantages of such a manufacturing sequence offset the delay and cost entailed in a double-machining setup. Warpage of thin sections during quenching is also a problem. Even in the same load, symmetry of cooling usually varies significantly among identical parts, and the resultant inconsistent warpage usually requires costly hand straightening. Consequently, a significant amount of effort has been devoted to reducing or eliminating warpage by changing racking position to achieve symmetry of cooling. For sheet metal parts, one manufacturer uses a double-screen floor in the quenching rack to reduce the force of initial contact between water and parts. Others allow parts to free fall from rack to quench tank. Spacing and positioning on the rack are carefully controlled so that parts will enter the water with minimum impact. With this technique, water turbulence must be avoided, because it will often cause parts to float for a few seconds, greatly reducing their cooling rate. Because of the difficulties encountered with quenching in cold water, milder quenchants have been employed. Indiscriminate use of milder quenchants can have catastrophic effects; however, when their use is based on sound engineering judgment and metallurgical knowledge of the effect on the specific alloy, significant cost-savings or performance improvement can Quenching of Aluminum Alloys / 171 be realized. The most frequent advantage is the reduction in costly straightening operations and in resultant uncontrolled residual stresses. For example, one aircraft manufacturer uses waterspray and air-blast quenching for weldments and complete formed parts made from 6061, an alloy whose corrosion resistance is relatively insensitive to quenching rate. Straightening requirements are negligible, and through careful control of racking and quenchant flow, the decrease in mechanical properties is minimized, as shown by the data in Fig. 42. Another development for reducing straightening cost is quenching in water-polymer solutions. Quenching of formed sheet metal parts in aqueous solutions of PAG has significantly reduced the cost of straightening parts after quenching. Because of the trade-offs of tensile properties with residual stress, researchers have been developing methods of analysis that combine prediction of properties by quench-factor analysis and prediction of stresses from heat-transfer analyses and other considerations. One of these methods predicts that a cooling rate that is slow at the beginning, but continuously accelerates, can significantly reduce residual stresses while maintaining the same mechanical properties as those obtained by quenching in cold water (Ref 40). Racking Practices (Ref 1) Proper racking is a key factor in controlling distortion and the level of residual stresses induced during the quench. Racking involves proper orientation and spacing of parts in a manner to permit free access of the heating and quenching media to all surfaces of parts in all portions of the load. If warpage and residual-stress levels are to be minimized, parts must be supported in the furnace correctly and enter the quenching fluid properly at the optimum rate. Improper racking and spacing can lead to low properties, spotty hardness, part melting, poor quenching response, excessive distortion, parts not reaching the proper temperature, and high residual stresses. Although proper racking to minimize distortion and residual stresses is a very complex subject that takes time and experience to master, proper racking of parts prior to the solution heat treating operation is probably the single most effective tool at the disposal of an aluminum heat treater for controlling distortion, both in the furnace and during the quench. The critical factors to consider when racking parts for solution heat treating include: Elevated-temperature strength characteris- tics: When aluminum alloys are heated to high temperatures, the elevated-temperature strength is extremely low and the material becomes very soft and pliable. Thermal expansion: Aluminum alloys have a high coefficient of expansion and will grow as the part is heated to the solution heat treat temperature. Part support: The part must be supported properly and not allowed to sag or bow during the heat and cooling operations. Part spacing: Parts must be spaced correctly, considering the thickness of the part and allowing sufficient air flow and quenchant to pass over the part to ensure even heating and cooling. Part configuration: Consideration must be given to the variation of thickness within a part that can lead to large differences in cooling rates, which cause excessive distortion or high levels of residual stress. Furnace design in relation to part orientation: Parts must be oriented in the furnace so that the air flow passes evenly over all surfaces of the part. Weight/configuration distribution: The size and weight distribution for the part and its relationship to the basket, rack, or fixture in which it is placed will have an effect on warpage. Condition of the baskets, racks, or fixture: Distorted baskets, racks, and fixtures make distorted parts. When considering how to rack parts in a particular furnace or oven, the operator must understand how the air flow travels so that in loading the parts, the air flow is not restricted, which can cause cold spots in the furnace. For example, if the air flow is from side to side, sheets of a large planer area must be positioned lengthwise with the flow so that the air is not restricted as it passes over all surfaces. Softness of Aluminum at Solution Temperatures. At solution heat treating temperature, the aluminum alloy is soft and will not hold its own weight. If hanging parts, especially where strength is important, steel wire should be used, not aluminum wire. For positioning parts, aluminum wire should be used, because it will allow the part to expand. Thermal Expansion. Aluminum alloys have a high coefficient of expansion and will grow as the part is heated to solution heat treat temperature. The degree of expansion and contraction during heating and cooling of aluminum parts is of critical importance. Table 14 illustrates the calculated expansion of a number of aluminum parts during heating from room temperature to a furnace temperature of 475 C (890 F). It can be seen that a 4.88 m (16 ft) long part is going to grow almost 5 cm (2 in.) in the furnace while it is at temperature. At room temperature, it may appear that there is sufficient spacing between the parts. However, as the temperature of the load is raised in the furnace, the expanding parts (if spaced too closely) will exert pressure on each other as they attempt to expand. Because the yield strength of the aluminum alloy is extremely low at elevated temperature, the parts Fig. 42 Effect of quenching medium on strength of 6061-T6 sheet. Water-immersion quench equals 100%. Control of coolant flow will minimize the decrease in mechanical properties. Table 14 Typical expansion of aluminum during solution treatment Part length at room temperature, 20 C (70 F) Expansion at 475 C (890 F) m ft mm in. 2.4 3.65 4.88 8 12 16 24 36 48 0.96 1.42 1.89 relieve the contact pressure by moving or bending to conform to the space available. When the parts are quenched, the distortion that has occurred in the furnace will not change; in many instances, it is erroneously concluded that the parts have warped during the quench. Unequal Expansion and Contraction. During the racking of a part, the problem of differential contraction between the part and the supporting baskets and fixtures must be considered. If a 2.4 m (8 ft) long part were to be quenched, a sudden contraction back to its original dimension would occur, and the part would shrink 25 mm (1 in.). If the part is not 172 / Heat Treating of Aluminum and Its Alloys allowed to expand or contract uniformly, without restriction, significant residual stresses or distortion will result during the quenching procedure. Because of these expansion characteristics, the use of fixtures for elevated-temperature processing operations (forming, welding, heat treating) must be carefully planned so that large differentials between the expansion characteristics of the part and its fixtures do not impart high levels of residual stress to the part, leading to dimensional instability. When quenching thin-gaged aluminum parts, the entire cooling process is completed in a matter of seconds or less, so the contraction of the part is almost instantaneous. Restricting the parts movement during the contraction is a major cause of distortion. If the parts are positioned by pins or bolts in a fixture, slotted holes are an absolute necessity. Also, the length and position of the slots should be computed, allowing for differential expansion of the part and the fixture. Centering the pin in the slot also may cause problems, because the direction of the expansion and contraction will not be based on the central location. Clamping the part too tightly may even cause dings in the part as the part expands into the clamp during heating. Proper racking of aluminum alloy parts to minimize distortion and residual stresses in the heat treating process is a very complex subject and takes time and experience to master. Not all of the various intricacies of racking can be covered in these few pages. However, by discussing a few of the basic principles and the effects racking can have on distortion, the reader may be convinced of the importance of learning and practicing good racking techniques. Effect of Racking Orientation on Part Warpage. The level and type of warpage is particularly pertinent when one considers different racking orientations, such as vertical, horizontal, or even angle racking. Parts that are laid flat in a basket usually result in the part bowing, while the same parts racked in a vertical position will normally twist if they have sufficient mass. Thin-gage sheet parts may bow, with the greater distortion being on the top of the part that was the last to enter the quenchant. Check and straightening time and costs also can be considerably different. Using basic principles, much quenching distortion can be eliminated or at least minimized. However, in spite of all possible efforts, many times, check and straightening operations are required to bring quenched aluminum parts back to within dimensional tolerance. If this situation occurs, if every part warps or distorts in the same manner, straightening costs can be significantly reduced. Thus, achieving the goal of what is called controlled warpage becomes important. This involves the principle that if all parts are spaced, racked, and quenched in the same manner, they will warp the same way. Controlled warpage makes it easier to straighten, because every part is the same. Allowance for Part Expansion and Contraction. If parts are restricted in any manner and not allowed to grow freely, the restriction will cause the part to distort. This distortion is frequently encountered when parts are tied too tightly to steel baskets or racks that are used to position the parts in the furnace. It also occurs when parts are racked too close together. At room temperature, it may appear that there is sufficient spacing between the parts. As the temperature of the load is raised in the furnace, the expanding parts (if spaced too closely) will exert pressure on each other as they attempt to expand. Because the yield strength of the aluminum alloy is extremely low at elevated temperature, the parts relieve the contact pressure by moving or bending to conform to the space available. When the parts are quenched, the distortion that has occurred in the furnace will not change; in many instances, it is erroneously concluded that the parts have warped during the quench. Restriction by racks, bars, or wires used to position the parts during the heat treating operation may cause a similar effect if the holding device is not sufficiently flexible to allow for the part to expand and contract freely. Any device used for positioning parts must be sufficiently loose to allow for thermal expansion and contraction. Parts Do not Move during the Quench. Parts should be tied lightly at the bottom or positioned appropriately with screen or other acceptable material at the top to ensure correct entry into the quenchant and to make sure they are secured and do not sail or move during the quench. The selection of a proper immersion rate is dependent on the part configuration; for many configurations, a slower immersion rate is appropriate. For some part configurations, Fig. 43 no matter how the part is racked, there will always be a large surface that offers resistance to the quenchant. In these cases, a slower immersion rate should be used. Thickness Variations and Heat Sinks. Thickness variations, such as encountered in forged and machined parts, may cause distortion during the quench because thin sections cool faster than thick sections. When racking parts with large thickness differences, the operator can compensate for thickness variations to some degree by his racking technique. A typical example is a machined part made from a 25 mm (1 in.) thick hand forging with machined pockets and a rib through the center with a section, such as shown in Fig. 43. The base of the pockets is machined down to 3.2 mm (1/8 in.) (before heat treating) with a 13 mm (½ in.) machined rib between the pockets. The part would be racked in the heat treat basket using small 6.4 to 9.5 mm (¼ to 3/8 in.) diameter steel rods to support the part vertically. Smooth, round rods made from alloy 4130 or 304 stainless are normally used. Several support rods placed along the thinner sections of the part will compensate for the variations in thickness (Fig. 43). The presence of these rods will reduce the cooling rate in the thin sections, keeping it closer to the rate in the thicker sections and thereby eliminating the canning effect that was experienced before the rods were used. Use of Heat Sinks. Many warpage problems that result from large thickness variations can be solved by the use of heat sinks. The term heat sink, although commonly used in the heat treating industry, is really a misnomer because these components, when added to the part, are not really sinks; they are intended to be heat sources. The purpose of the heat sink is to increase the Thickness variations of machined part being quenched, with location of racking bars along the thin bottom to act as a heat sink. Source: Ref 1 Quenching of Aluminum Alloys / 173 mass of the part in a specific area so that more equalized cooling is achieved throughout the entire part. Increasing the thickness or mass in a thin area allows that area to cool at the same rate as thicker areas, thereby reducing the warpage during the cooling process. The use of heat sinks must be done carefully. It must be performed in a manner so that the area of the part is not cooled so much that it affects the final heat treat properties. Inspection of the affected area must be rigidly controlled. In some instances, application of quench-sensitivity data can be helpful in determining the type and size of the method used. Also, careful adherence to specification requirements must be practiced. Some prime contractors do not allow the use of heat sinks without prior approval. Heat sinks take on different forms. It is common practice to use aluminum screen, sometimes bunched in a ball or wrapped around an aluminum block, to act as a heat sink. This technique has proven to be effective in controlling differential quenching rates in many cases. To compensate for severe canning distortion in the machined pockets of a large wing spar, another example involved the use of cast aluminum waffle plates, which were positioned in the bottom of each pocket to equalize the cooling rates with the surrounding ribs. The waffle plates were fastened to the part using bolt through holes in each pocket, and they completely eliminated the canning of the pocket areas. Spacing of Parts. As previously noted, the fundamental requirement of proper racking is that “all parts be racked or supported in a manner to permit free access of the heating and quenching media to all surfaces of parts in all portions of the load.” This requirement establishes that parts must be spaced properly to ensure the free access of the heating and quenching media. Most specifications have this requirement in some way or another. Unfortunately, there is a wide difference of opinion among most experts regarding the exact spacing that is needed to achieve this goal. Specification requirements vary significantly regarding the exact spacing that is needed. Some companies leave the spacing requirement up to the heat treater as long as the primary requirement is met. The first requirement of racking for the quench is to allow sufficient space around each part so the quench medium has free access to all surfaces. This spacing is essential so that the fluid can effectively extract heat from all surfaces uniformly throughout the entire quench. Allowing the quenchant to become too hot in any location near the part because of improper spacing or inadequate agitation can lead to distortion and lower mechanical properties. Problems often occur while quenching some of the bulkier forgings and castings. At first appearance, because of their size and thickness, distortion would not appear to be a problem. To obtain the most production from the available equipment, the heat treater may stack or simply dump parts into the basket. When the parts are quenched, the lack of proper spacing does not allow free access of the quenching fluid. When only small amounts of fluid (usually water) reach between parts, local steam or vapor pockets develop. The presence of these vapor pockets slows down the cooling rate at these locations. Slower cooling rates allow the alloying elements to come out of solid solution, and the loss of these alloying constituents can result in a significant loss of hardness and strength properties after aging. The reduced properties are usually observed in the parts during hardness testing and normally appear as soft spots. In some cases, entire parts will be below the minimum hardness and strength requirements for that alloy and temper. Distortion or the inducement of unwanted residual stresses can easily occur in heavier parts when inadequate or uneven spacing is used. Spacing should increase with the part thickness, because it will require a larger volume of quenchant between parts to extract the greater amount of heat contained in the thicker part. When quenching in water, the spacing between parts should be increased as the water temperature is increased. The quenching power of water decreases as it is heated. Water temperatures above 70 C (160 F) exhibit very slow cooling rates and should not be used for most of the thicker parts unless allowances are made for reduced properties. Quench Tank Systems Immersion in tanks, frequently of unlined waterproof concrete, particularly if they are below floor level, is the most usual method. Small tanks and those above floor level usually are metal. Aluminum or stainless steel is preferred for metal tanks. If carbon steel is used, suitable surface coatings must be applied to suppress corrosion, which can cause staining of the product. Water used for quenching also may contain a small percentage (usually less than 1 to 2%) of a rust inhibitor to minimize rusting of the quench tank and racks. Quench tanks must be large enough to permit the furnace charge to be immersed completely, preferably below the excessively heated top layer of water. For quenches specified at room temperature, water volume should be sufficient to maintain the bath below 40 C (100 F) during the quenching cycle. If this is not practical, bath temperature must be kept below this maximum through vigorous agitation by such means as high-velocity introduction of makeup water or recirculation with pumps or propeller-type mixers. When slower quench rates are required, the quenching medium may be kept above normal ambient temperature by adding suitable heating coils or steam injection facilities to the tank. Uniform quench bath temperature is essential to optimum product characteristics. The practical aspects and design considerations of a quench system involve a number of variables that include alloy type, product type, load configuration, quenchant fluid, agitation rate, total load weight, and the density of the load on the racks. The focus of this section is on design considerations for immersion tank systems in terms of: Batch or continuous process Materials selection Heat load Quench tank and rinse tank agitation Part tacking and baskets Cooling/heating Fluid maintenance Concentration control and separation methods Some practical aspects of quench system design depend on product type. For example, quench systems are typically divided according to product gage: one for sheet metal parts and parts up to 6 mm (¼ in.) thickness, or a system for heavier-gage parts that have cross sections more than 6 mm (¼ in.) thick. The main difference between sheet metal quenching and a heavy-gage load is the fact that sheet metal will normally be cooled by the time the parts reach the bottom of the quench bath. The hoist or elevator provides the main means of agitation for the cooling cycle, while the agitator system only needs to provide proper mixing and uniformity of the tank before and during the quench. Batch or Continuous Process. Quench tanks will support either a batch process or a continuous process. Batch quenching is used for furnaces that use an indexing system to present a load for quenching in a tank sized with enough volume of quench that the temperature rise in the tank is within acceptable limits for each quench cycle. Other types of furnaces are horizontal or vertical quench furnaces (drop-bottom furnaces). Continuous quench tanks are sized with enough volume of liquid to allow for proper agitation and heat removal from the work as it is quenched. This, in connection with a properly sized cooling system, ensures the process can continue without exceeding the upper limit of quench temperature. Several different methods are used to provide proper flow and cooling for the parts. There are chute quench systems, spray quenching, and simple tanks with a belt. Continuous quench tanks are mostly employed in connection with belt furnaces. Other types of furnaces are walking beam furnaces or screw furnaces. The continuous quench tanks accommodate both bulk products and single-piece product quenching. Tank Materials Selection. Aluminum can be affected by free iron (rust) in the quench bath during the quenching. Surface corrosion is particularly troublesome with sheet metal parts. The corrosion will show up as dark splotches that, with closer examination, reveal a black spot in the center (iron particle). Free iron is not the 174 / Heat Treating of Aluminum and Its Alloys only cause for surface corrosion but can be a contributor. Other contributors can be contamination of the parts by oil, cutting fluids, and poor materials handling before entry to the heat treat. For forging and castings, there are normally no problems because secondary machining or surface treatment removes the condition. The main source for the free iron is the tank wall and agitation system if the tank is made from mild steel. Secondary contributors can be heat treating racks and fixtures and finally the piping materials used for agitation and pumping. With the aforementioned in mind, the tanks for heavy castings and forgings are normally made from mild steel, with stainless baffles, agitators, and elevators. The tanks for sheet metal parts are mostly made from stainless steel, with all internal components made from stainless steel. Most of the piping is made from chlorinated polyvinyl chloride (CPVC) or stainless steel for water and hot water quench tanks. For PAG tanks, the shell and components can be made from mild steel. This is due to the PAG corrosion inhibitor (sodium nitrate), which will protect the tanks and piping and thus the parts. Several coatings have been tried over the years, with various successes. The most successful are the two-component epoxy coatings. However, PAG and hot water have a tendency to lift any coating from the metal, especially where mechanical damage has occurred during operation. Coatings are normally used as a cost-saving measure compared to the use of stainless steel. The author does not recommend this practice because the cost of replacements and repairs to the coatings will exceed the initial cost of the stainless steel lining. Polyvinyl chloride and CPVC piping can be used if care is taken to protect the piping from the hot load and direct heat from the open Fig. 44 furnace. It must always be remembered that Murphy’s law will ensure the tank is exposed to the full heat from the furnace when it is stuck under a quick quench or when a hot basket becomes stuck on an elevator. Heat Load and Tank Sizing. In accordance with most of the aluminum specifications, the tank is sized so the temperature rise does not exceed 5.5 C (10 F) for parts processed in accordance with AMS 2770, “Heat Treatment of Wrought Aluminum Alloy Parts,” and AMS 2771, “Heat Treatment of Aluminum Alloy Castings.” Other industries allow a higher temperature rise for castings and forgings; typically, a temperature rise of 11 C (20 F) is allowed. As an example, Fig. 44 illustrates the heat load calculations for quenching 2300 kg (5000 lb) of aluminum parts placed in a 680 kg (1500 lb) steel rack. The temperature of the water quench is 70 C (160 F). The parts and rack are heated to 540 C (1000 F) in the furnace. As shown in the example, the tank volume must be a minimum of 50,515 L (13,345 gal) of water to ensure that the quench temperature does not rise more than the specified 5 C (10 F). Standard practices do not include the heat requirement for heating the tank shell and other components that come into direct contact with the quench. This provides an additional safety factor for temperature rise. This calculation should always be done to determine the minimum volume of the tank. In addition to the volume, the size of the tank must also accommodate the parts and rack being processed. Sufficient clearance is needed for the instrumentation, agitation, and maintenance access to the components. The next step in the process is to consider the required agitation rate for the type of product processed in the quench facility. Heat load calculations for sizing of a quench tank in terms of British thermal units (Btu), where 1 Btu = 1054 J. This example is based on quenching 2300 kg (5000 lb) of aluminum parts placed in a 680 kg (1500 lb) steel rack. The parts and rack are heated to 540 C (1000 F) in a furnace. The temperature of the water quench is 70 C (160 F), and the allowable temperature rise is 5 C (10 F). See text for additional discussion. Quench Tank Agitation. There have been various ways of specifying agitation in a design, such as: Changeover of tank volumes (gallons per hour) Description of surface movement (babbling brook) Measured flow past the parts (feet/seconds) The best way to specify the quench flow is a calculated or measured flow past the parts. The maximum flow that should be specified for aluminum batch quenching with water or PAG solution is on the order of 24 to 36 cm/s (0.8 to 1.2 ft/s) past the parts. Any flow higher than this will not add to the cooling of the parts unless spray quenching is used. This maximum rate of fluid may not be practical in large tanks and would mean the complete tank volume must be changed over every 1 to 3 min. Many tanks successfully produce heavy-gage parts with measured flows on the order of 7 to 12 cm/s (0.25 to 0.4 ft/s). The following are basic guidelines for flows in quench tanks that have proven acceptable: Part thickness Product Thin sheet metal Heavy sheet metal Plate and machined parts mm in. Flow rate cm/s ft/s <2.3 <0.090 3–9 0.1–0.3 2.3–6 0.090–0.25 9–24 0.3–0.8 Up to 75 Up to 3 15–30 0.5–1.0 Agitation for flow rates more than 36 cm/s (1.2 ft/s) past the parts will not improve the cooling rate for water and polymer quench. As previously noted, the main difference between quenching sheet metal (up to 6 mm, or ¼ in., thick) and a load of heavy-gage parts (more than 6 mm, or ¼ in., thick) is the fact that sheet metal will normally be cooled by the time the part reaches the bottom of the quench bath. The hoist or elevator provides the main means of agitation for the cooling cycle. As a general rule for sheet metal parts, the hoist speed should be as slow as possible to avoid high hydraulic forces on the soft metal. The quench must still be completed within the allowable quench delay for the type of metal and furnace used. A slower speed will reduce distortion of the part. To obtain a slow elevator speed, the travel distance from furnace to quench must be as short as possible. New drop-bottom furnaces with moveable quench carts are superior designs compared to older pit-type drop-bottom furnaces in this regard. For heavy parts, agitation has a much more important role in removing heat from the parts. Practical experience and research by others has shown that a quench flow of approximately 0.3 m/s (0.8 to 1.2 ft/s) is adequate to generate the required cooling on heavy-gage parts. Faster water flow does not increase the cooling rate significantly, and high flow rates increase the difficulty of design criteria and require higher Quenching of Aluminum Alloys / 175 power consumption for the agitation. See the section “Agitation Systems” in this article for more information. When dealing with quenchants, it must be noted that direct high-velocity impingement of the fluid against the part must be avoided to ensure that spot cooling does not occur. Spot cooling can cause severe distortion and uneven properties in the finished product. It also is important to realize that quench agitation is different than mixing of chemicals. Heat treat facilities are specifically looking for the linear flow with some turbulence past the part that gives the best and most efficient cooling of the part in a predictable manner across the whole section of the product rack or part each time a quench is performed. Flow patterns and the extent of flow depend on how the tanks are configured with the agitation. Tanks can be divided in halves or thirds, depending on how the agitation is designed, to optimize the flow pattern effect (Fig. 45). Tank designs must take these natural flow patterns into account to optimize agitation in the load with minimum power and energy consumption. However, in many cases, space constraints at the facility create a need to optimize the assigned space and load size while still generating sufficient flow around the parts to ensure good properties after the quench-and-age cycle. Water Heating and Cooling. The quench tank must be equipped with a means of initial heatup if the tank is used for quenching in hot water. For temperatures above 70 C (160 F), a precaution is to insulate tanks and piping to protect personnel. In addition, the insulation reduces heat losses during slow production. Some areas of the country have very hard water, and calcium deposits on heating elements can cause damage. The heating can be done with steam, natural gas, or electric. The most commonly used heating medium is a submerged burner tube fired by natural gas or electric heating elements submerged directly in the tank. Flow-through electric heaters are also used. A heat-up time of 6 to 8 h is normally used. During production, the parts that are quenched provide the heat. The control for the heating is an on/off system. There are no requirements for proportionalintegral-derivative control due to the very slow response time of the tanks. The agitation must be a well-integrated design with the heaters to ensure that there is good flow across the heaters and that temperature uniformity is achieved in the tank during heatup. Cooling of the tanks is done by the use of heat exchangers or a chiller. The heat exchangers can be water/water or water/air. Water/air exchangers are placed either inside or outside of the buildings. The sizing of the cooling systems will depend on how fast the tank is required to recover to the start temperature. Part Racking and Baskets. The rack must be fabricated of materials that can endure repeated heating and cooling cycles without any detrimental effect on the rack, as described in the section “Racking Practices” in this article. The racks must be pinned and bolted together to allow the rack to expand and contract without restriction during heating and cooling. Welding must be eliminated as much as possible, because racks have a high tendency to crack. The use of tubing, especially 4130 steel tubing, has been very successful throughout the aluminum aerospace industry with racks that heat to a maximum of 565 C (1050 F). These production racks have thousands of cycles without any repairs or distortion. For temperatures above 565 C (1050 F), other material is required. The round tube or rod shape is preferred to structural shapes such as I-beams or C-channels, which will not cool evenly during the quench and will distort severely after a few quenches. Load configuration is probably the most important aspect of heat treating heavy-gage loads. The load must be configured to allow the air to heat the parts during the heat cycle, and the parts must be spaced so the quench has access to all surfaces and can remove the heat quickly. The tendency to pack same-sized and -configured parts tightly on the racks can have a very detrimental effect on the process. The tightly packed parts can have significantly different properties after heat treating than the same parts spaced properly on the fixture. In fact, this links back to the problem with tank modeling and parts testing for the design of quench tanks. If single parts or a small load are used for this testing, the results can be different than the actual production loads. The rule of thumb is that there must be a minimum of 25 mm (1 in.) plus the thickest part of the material between each part to achieve good heat transfer. Care must be taken when racking the parts. As shown in Fig. 46, it can significantly change the process when different approaches are used for hanging the same part. The main concern in the example shown in Fig. 46 is the fact that the steel rod has a different cool-down rate than the aluminum, and the part may have a soft spot where the rod is in contact with the part due to slow cooling and slow heatup during the solution heat treat cycle. The rod prevents proper access and cooling for the quench. The use of thin-walled tubing/pipe for hanging the parts is preferred compared to solid rod. Fluid Maintenance and Control. In addition to tank design and the proper degree of agitation, the cleanliness of the quenchant is an important factor in the quench system. A dirty, contaminated quench bath can have significantly different quench qualities and cooling capabilities than a clean bath. Contamination can be categorized as particle contamination, chemical contamination, and biological contamination. Particle contamination can come from several sources: tank and rack scale, sand and dirt from the factory environment, or from manufacturing of the part itself. One of the release agents used in the forging process is graphite, which adheres to the parts and is washed off in the quench. Sand from the casting mold is another example. Filters are employed to remove dirt from the tank to maintain a reasonably clean quench. The filters must be sized to allow for maintenance. If they are too small, the changing/cleaning will be too big a burden; if they are too large, equipment cost is a factor. It is important to obtain an estimate of the dirt loading in the tank before the design is decided for the filtering system. Bag filters or cartridge filters are the most commonly used filter types. Filter sizes are in the 5 to 10 mm range. For sand removal, centrifuge-type filters are used along with conveyor systems for the heavier loadings. Chemical Composition Control. The use of PAG polymers and additives involves some Fig. 45 Basic types of flow patterns in quench tanks. Load baskets are outlined, and shaded areas are regions of almost zero flow. (a) Tank is divided into three flow areas, with central location of flow from sparger pipes. (b) Flow pattern of tank with side agitator (draft tube) Fig. 46 Use of tube for hanging parts. The use of thinwalled tubing/pipe for hanging the parts is preferred, because solid rod prevents proper access and cooling for the quench. 176 / Heat Treating of Aluminum and Its Alloys additional requirements on composition control (see also the section “Polymer Quenchants” in this article). The PAG-water solution quenchants consist of polymers and several different additives. The polymer molecule does not change much during the life of the bath, which can be several years in a properly maintained system. However, some of the components can disappear over time. The corrosion inhibitor (sodium nitrate) can be diluted and removed with some concentration methods, and the pH level can change. Low pH levels can damage parts by an etching effect. The heat treating facility must implement a regimented qualityassurance program that will detect problems in the quench before they become detrimental to the process. The supplier of the polymers can assist in testing and replenishments of the chemicals as needed to maintain the bath. Concentration measurement for PAG quenchants includes densitometers, refractometers, and viscosity meters. The cleanliness of the bath directly influences the accuracy of the measurements. Several of the instruments require frequent calibration. The electronic refractive index monitor with remote sensing and optional connection to a PLC has proven to be very stable if the solution is conditioned and filtered. Accuracy levels can be within ±0.5% over time, with only very limited maintenance requirements. Bioburden Control. Bacteria and algae growth can occur in aqueous solutions if there are no biocides present. Biocides are used with various successes to control the problem. Biocides with glutealdehyde are the most commonly used. They last from 10 to 21 days in the bath and must be replenished periodically to remain effective. The amount varies according to contamination levels. Approximately 150 to 250 parts per million added every two weeks can normally keep the bath in complete control. It is highly recommended to use an automatic injection system to limit worker exposure to the very toxic materials used in biocides. Shop test procedures check for bacteria and fungi. An occasional change of biocide will keep the bacteria from becoming resistant to the product. Agitation Systems Flow is generated by using several methods. Quench tanks used in the aluminum industry can contain very large quantities of water (38,000 to 227,000 L, or 10,000 to 60,000 gal), and to move this amount of water at an average speed of 0.3 m/s (1 ft/s) requires significant pumping. There are mainly two options in these cases: pumping with water-jet eductors at the sparging pipes or agitators in draft tubes. Pumping and the use of different types of propeller agitation provide the most common method. Parts or basket movement are used in rare occasions. Pumping is versatile and does not take up much space in the tank, because sparger pipes, eductors, and nozzles can be tucked close to Table 15 Comparison of power the sidewall or bottom of the tank. Pumping requirements for draft tubes and pumping has a low efficiency per gallon of quench systems moved compared to other types of agitation Pumping systems devices, especially draft tube designs. The use Draft tubes (end suction pumps) of an eductor can significantly increase the Flow rate, rpm Propeller type hp psi hp amount of quench moved inside the tank. gal/min 5600 810 13.5 in. airfoil 5.5 20 75.5 The volume increases by a factor of 4 and 520 13.5 in. airfoil 2.0 20 42.6 the velocity decreases by the same factor. 3200 2950 426 13.5 in. airfoil 1.0 20 39.0 However, the overall flow generated will be sufficient to make a good quench. Compared to nozzles, an eductor provides a better distribution of the flow and does not generate point- Top entering Elevator cooling of parts by hitting the part with a very agitator or basket high velocity of fluid at a concentrated spot. guides Propeller agitation is divided between open placement and agitation tube placement. In Fluid level addition, there are marine-type propellers and airfoil-type propellers used for agitation purOverflow weir poses. The open-type propellers are most commonly used in side-mounted systems, for example, integral quench furnaces. These proPropeller pellers are typically marine-type propellers, which are slow spinning compared to airfoilGrating or Draft type propellers. The swirling action of the perforated plate tube quench when it leaves the propeller tips generates a good nonlinear flow. However, the flow Directional flow baffles is very uneven and can affect properties in the parts. The horsepower requirements are large compared to airfoil-type systems; however, it Fig. 47 Quench tank with single draft tube agitator is less than pumping. Table 15 shows a comparison of required power between pumping and for quench tank and furnace design is used to draft tubes. The draft tube is widely used in the larger verify and predict the mechanical design and open-tank systems. Draft tube design is covered process variables. With the new programs, it in detail in Ref 13. Basically, the draft tube is possible to provide easy-to-understand graconsists of a propeller (airfoil or marine type) phics that can be used in the design decisions placed inside a tube (Fig. 47). The placement for different quench tanks. With modeling proof the propeller inside the tube increases the grams, it is possible to review and change paraefficiency of the propeller and gives the meters and then observe the calculated results. Parts testing is typically employed when designer the ability to direct the quench flow in a more controlled and predictable manner. existing equipment is used for new products The distance from the water to the edge of the or for improvements of existing product proflared tube must be large enough to prevent cesses. A proper test plan must be developed air from being pulled down into the tube and that addresses the areas that can affect the part: thus creating bubbles in the quench. The bub- placement of the part in the tank, orientation of bles can create an insulating layer on the the part in the tank, and areas of high and low parts and must be avoided in the quench tank. flow in the tank. It is very important to find Several methods are available to prevent the out where the flow is in the tank by mapping vortex from being started. One is to place a flat it with a flow meter. An open-type flow meter plate 5 cm (2 in.) under the surface and force is preferred to a closed-type flow meter, as the water to enter the agitator in a more shown in Fig. 48. The measurements of the flow will normally be horizontal manner. This will create a slight restriction in the inlet, but normally this will taken without parts in the tank. When the parts not reduce the volume significantly. The other displace space and volume in the tank, the speed method is to place the propeller and the flared of the quench around the parts increases. In addicone deep enough to prevent the inlet vortex tion, the thermal action of the rising heated quench from contact with the parts adds to the from forming. Flow modeling and parts tests are useful velocity of the quench past the parts. With this in tools in evaluating the proper method for gener- mind, it is understandable that tanks with lessating the flow and placing parts baskets in the than-desired flows that empty as described in this area of maximum flow. The use of mechanical article can, in fact, produce satisfactory parts. Example 1: Correction of Uneven Flow. tank modeling can help design an agitation system, but building of models can be time-con- A 57,000 L (15,000 gal) quench tank was agisuming, and scaling from the model to the tated by three large side-mounted marine-type finished tank size and product may not be a propellers. The quench area for the parts was straightforward process. Computer modeling in the top 41 cm (16 in.) of the tank, because Quenching of Aluminum Alloys / 177 parts were quenched one at a time every 20 to 30 s. The flow was very strong but uneven, as shown in Fig. 49. Several methods were used to solve the problem. Baffling and flowdirection vanes did very little to even out the flow. The final fix was to install a perforated plate under the parts. The perforated plate/plenum created a very even and desirable flow. Figure 50 shows the surface of the tank after installation of the plenum. A crown of approximately 50 mm (2 in.) can be seen in the middle of the tank where the quench is forced up and then returns to each side of the tank. The use of perforated plenums in conjunction with tube or open-type agitators is very successful in generating controlled, even flows. Fig. 48 Fig. 49 ACKNOWLEDGMENT Tom Croucher, who was widely known in the U.S. aerospace industry as an expert in heat treatment, passed away while this article was being developed. With sadness in the loss of Tom, the editors want to recognize his contributions and expertise in quenching, distortion control, polymer quenching, and the application of uphill quenching process. Tom is missed. REFERENCES 1. T. Croucher, Fundamentals of Quenching Aluminum Alloys, May 2014 Difference between closed flow meter and open propeller flow meter Uneven flow in quench tank. Courtesy of Bogh Industries Fig. 50 2. W.L. Fink and L.A. Willey, Quenching of 75S Aluminum Alloy, Trans. AIME, Vol 175, 1948, p 414–427 3. J.W. Evancho and J.T. Staley, Kinetics of Precipitation in Aluminum Alloys during Continuous Cooling, Metall. Trans. A, Vol 5, Jan 1974, p 43–47 4. J.W. Evancho, “Effects of Quenching on Strength and Toughness of 6351 Extrusions,” Report 13-73-HQ40, Alcoa Laboratories, 1973 5. J.T. Staley, Quench Factor Analysis of Aluminium Alloys, Mater. Sci. Technol., Vol 3, Nov 1987, p 923–935 6. T. Sheppard, Mater. Sci. Technol., Vol 4, July 1988, p 636 7. D.V. Gullotti, J. Crane, and W.C. Seber, Isothermal Transformation Characteristics of Several 6xxx Series Alloys, Proc. Second International Aluminum Extrusion Technology Seminar, Vol 1, Billet and Extrusion, Nov 15–17, 1977 (Atlanta), p 249–256 8. Heat Treating, Cleaning and Finishing, Vol 2, Metals Handbook, 8th ed., American Society for Metals, 1964 9. W.A. Anderson, Precipitation from Solid Solution, American Society for Metals, 1958, p 167 10. H.Y. Hunsicker, The Metallurgy of Heat Treatment, Aluminum: Properties, Physical Metallurgy, and Phase Diagrams, Vol 1, D.R. Van Horn, Ed., American Society for Metals, 1967, p 109 11. D.O. Sprowls and R.H. Brown, Alcoa Technical Paper 17, 1962 12. Aerospace Structural Materials Handbook, Purdue Research Foundation, 1992 More uniform flow compared to Fig. 49. Note crown compared to outside. Courtesy of Bogh Industries 178 / Heat Treating of Aluminum and Its Alloys 13. G. Totten, C. Bates, and N. Clinton, Handbook of Quenchants and Quenching Technology, ASM International, 1993 14. J.E. Hatch, Ed., Aluminum Properties and Physical Metallurgy, American Society for Metals, Metals Park, OH, 1983 15. C.E. Bates, Selecting Quenchants to Maximize Tensile Properties and Minimize Distortion in Aluminum Parts, J. Heat Treat., Vol 5 (No. 1), 1987, p 27–40 16. K. Speith and H. Lange, Mitt. Kaiser Wilhelm Inst. Eisenforssch, Vol 17, 1935, p 175 17. A. Rose, Arch. Eisenhullennes, Vol 13, 1940, p 345 18. C.E. Bates, G.E. Totten, and R.J. Brenner, Heat Treating, Vol 4, ASM Handbook, ASM International, 1991, p 51 19. “Heat Treatment of Wrought Aluminum Alloys,” AMS 2770, SAE International, Warrendale, PA 20. T. Croucher and M.D. Schuler, Met. Eng. Q., Aug 1970 21. R.H. Lauderdale, “Evaluation of Quenching Media for Aluminum Alloys,” MDR 6-18002, Boeing, March 1967 22. E.A. Lauchner and B.O. Smith, “Evaluation of UconW Quenching,” NOR 69-65, Northrop Corporation, May 1969 23. T.R. Croucher, Applying Synthetic Quenchants to High Strength Alloy Heat Treatment, Met. Eng. Q., May 1971 24. T.R. Croucher, Synthetic Quenchants Eliminate Distortion, Met. Prog., Nov 1973 25. S. Chaudhury and D. Apelian, Fluidized Bed Heat Treatment of Cast Al Alloys, Proc. John Campbell Symposium, TMS Annual Meeting (California), 2005, p 283 26. S. Chaudhury and D. Apelian, Effect of Rapid Heating on Solutionizing Characteristics of Al-Si-Mg Alloy Using a Fluidized Bed, Metall. Mater. Trans. A, Vol 37, 2006, p 763–778 27. J. Keist, D. Dingmann, and C. Bergman, Fluidized Bed Quenching: Reducing Residual Stresses and Distortion, Proc. 23rd Heat Treating Society Conference (Pittsburgh, PA), 2005, p 263–270 28. M. Avrami, Kinetics of Phase Change I, J. Chem. Phys., Vol 7, Feb 1939, p 1103–1112 29. M. Avrami, Kinetics of Phase Change II, J. Chem. Phys., Vol 8, Feb 1940, p 212–224 30. J.W. Cahn, Acta Metall., Vol 4, 1956, p 449–459 31. R.J. Flynn and J.S. Robinson, The Application of Advances in Quench Factor Analysis Property Prediction to the Heat Treatment of 7010 Aluminium Alloy, J. Mater. Process. Technol., Vol 153–154, 2004, p 674–680 32. G.P. Dolan and J.S. Robinson, Residual Stress Reduction in 7175-T73, 6061-T6, and 2017A-T4 Aluminum Alloys Using Quench Factor Analysis, J. Mater. Process. Technol., Vol 153–154, 2004, p 346–351 33. G.P. Dolan, J.S. Robinson, and A.J. Morris, Quench Factors and Residual Stress 34. 35. 36. 37. 38. 39. 40. Reduction in 7175-T73 Plate, Proc. Materials Solution Conference (Indianapolis, IN), ASM International, 2001, p 213–218 D.D. Hall and I. Mudawar, Optimization of Quench History of Aluminum Parts for Superior Mechanical Properties, Int. J. Heat Mass Transf., Vol 39 (No. 1), 1996, p 81–95 L.K. Ives et al., “Processing/Microstructure/ Property Relationships in 2024 Aluminum Alloy Plates,” National Bureau of Standards Technical Report NBSIR 83-2669, U.S. Department of Commerce, Jan 1983 L. Swartzendruber et al., “Nondestructive Evaluation of Nonuniformities in 2219 Aluminum Alloy Plate—Relationship to Processing,” National Bureau of Standards Technical Report NBSIR 80-2069, U.S. Department of Commerce, Dec 1980 J. Newkirk and D. MacKenzie, The Jominy End Quench for Light-Weight Alloy Development, J. Mater. Eng. Perform., Vol 9 (No. 4), 2000, p 408–441 P.M. Kavalco, L.C.F. Canale, and G.E. Totten, Quenching of Aluminum Alloys: Property Prediction by Quench Factor Analysis, Heat Treat. Prog., May/June, 2009 K.R. Van Horn, J. Met., March 1953, p 405–422 P. Archambault et al., Heat Treatment 1976, Proc. 16th Int. Heat Treatment Conference, Book 181, Metals Society, London, 1976, p 105–109, 219, 220

![[ Graphics Card- 710-1-SL]](http://s2.studylib.es/store/data/005308161_1-3d44ecb8407a561d085071135c866b6c-300x300.png)